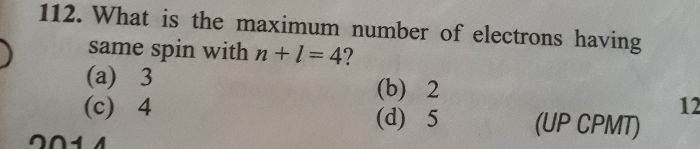CBSE Class 11-science Answered
Arrange the electrons represented by the following set of
quantum number in decreasing order of energy.
(i) n = 4, l =0, m=0 S= +
(ii) n = 2, l =1, m=1 S= -
(iii) n = 3, l =2, m=0 S= +
(iv) n = 3, l =0, m=0 S= -
Asked by Neetukhaiwal | 24 Jul, 2021, 11:18: AM
Order of energy is calculated by (n+l) rule. Orbital which is having lower (n+l) value will have lower energy and If values of (n+l) is same than orbital which is having lower value of n will have lower energy,
So, Decreasing order of energy-
(iii)>(i)>(iv)>(ii)
Answered by Ravi | 29 Jul, 2021, 11:10: AM
Concept Videos
CBSE 11-science - Chemistry
Asked by deba.biswas561 | 14 Jun, 2022, 08:07: AM
CBSE 11-science - Chemistry
Asked by Neetukhaiwal | 24 Jul, 2021, 11:18: AM
CBSE 11-science - Chemistry
Asked by salvadormiranda2509 | 13 Feb, 2021, 12:53: PM
CBSE 11-science - Chemistry
Asked by sulaikhasulu393 | 01 May, 2020, 16:37: PM
CBSE 11-science - Chemistry
Asked by prashantyadav592 | 29 Jan, 2020, 09:39: AM
CBSE 11-science - Chemistry
Asked by Debdulal | 29 Aug, 2019, 16:47: PM
CBSE 11-science - Chemistry
Asked by sairamsribaba80 | 28 Aug, 2019, 18:54: PM
CBSE 11-science - Chemistry
Asked by timeyashkadam | 19 Apr, 2019, 10:22: AM
CBSE 11-science - Chemistry
Asked by himankarbhati3 | 21 Sep, 2018, 08:29: AM
CBSE 11-science - Chemistry
Asked by ramandeep.kaur | 06 Sep, 2018, 13:21: PM