CBSE Class 11-science Answered
The volume of the gas at 0'C and 700mm pressure is 760 CC.The no.of molecules present in this volume is ?
Asked by minipkda | 18 Aug, 2018, 20:46: PM
Given:
Temperature T = 0 °C
= 273 K
Pressure P = 700 mm
1 atm = 760 mmHg
Therefore,
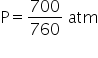
Volume V = 760 cc
= 760×10-3 litre

The 1.88×1022 molecules are present in the given volume of gas.
Answered by Varsha | 20 Aug, 2018, 12:08: PM
Concept Videos
CBSE 11-science - Chemistry
Asked by pratikshyadashrkl | 12 Apr, 2020, 18:47: PM
CBSE 11-science - Chemistry
Asked by Ankit | 16 Mar, 2019, 13:01: PM
CBSE 11-science - Chemistry
Asked by minipkda | 18 Aug, 2018, 20:46: PM
CBSE 11-science - Chemistry
Asked by smanishkumar2002 | 04 Aug, 2018, 05:36: AM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 20 Apr, 2015, 11:17: AM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 20 Apr, 2015, 11:21: AM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 20 Apr, 2015, 11:33: AM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 20 Apr, 2015, 11:32: AM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 20 Apr, 2015, 13:04: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 20 Apr, 2015, 11:38: AM






