CBSE Class 12-science Answered
alpha decay:
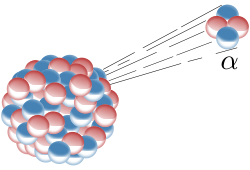
Alpha decay is a type of radioactive decay in which an atomic nucleus emits an alpha particle (two protons and two neutrons bound together into a particle identical to a helium nucleus) and transforms (or 'decays') into an atom with a mass number 4 less and atomic number 2 less. For example:
although this is typically written as:
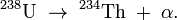 An alpha particle is the same as a helium-4 nucleus, and both mass number and atomic number are the same.
An alpha particle is the same as a helium-4 nucleus, and both mass number and atomic number are the same.
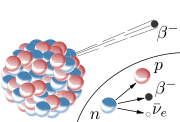
beta decay is a type of radioactive decay in which a beta particle (an electron or a positron) is emitted. In the case of electron emission, it is referred to as "beta minus" (β− ), while in the case of a positron emission as "beta plus" (β+ ).
Gamma rays are often produced alongside other forms of radiation such as alpha or beta. When a nucleus emits an α or β particle, the daughter nucleus is sometimes left in an excited state. It can then jump down to a lower level by emitting a gamma ray in much the same way that an atomic electron can jump to a lower level by emitting visible light or ultraviolet radiation.