CBSE Class 11-science Answered
what will be the answer

Asked by saranyachakraborty2007 | 25 Apr, 2024, 05:23: AM
Dear Student,
The minimum energy required to overcome the attractive forces between an electron and the surface of silver (Ag metal)
= 5.52 × 10-19 J
Wavelength λ = 360 Ao
Maximum Kinetic Energy of the electron ejected out from silver which is being exposed to UV light =?
Energy absorbed = hc / λ
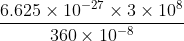
= 5.52 × 10-11 erg
or 5.5 × 10-18 J (1 erg = 10-7 J)
The energy of photon = Work function + Kinetic energy
So, Kinetic energy = (5.52 ×10-18 J) - (5.52 × 10-19 J) = 49.68 ×10-19 J
Answered by | 26 Apr, 2024, 10:23: AM
Concept Videos
CBSE 11-science - Chemistry
Asked by saranyachakraborty2007 | 25 Apr, 2024, 05:23: AM
CBSE 11-science - Chemistry
Asked by habibakhatoon112 | 15 Jul, 2022, 09:14: PM
CBSE 11-science - Chemistry
Asked by akankhyapradhan123 | 22 Oct, 2021, 07:29: PM
CBSE 11-science - Chemistry
Asked by kusumghosh71 | 21 Aug, 2021, 10:59: AM
CBSE 11-science - Chemistry
Asked by muramshettyaashrith218 | 16 Sep, 2020, 09:58: PM
CBSE 11-science - Chemistry
Asked by nandanappillai | 21 May, 2020, 01:08: PM
CBSE 11-science - Chemistry
Asked by rajarage18 | 23 Jan, 2020, 08:00: PM
CBSE 11-science - Chemistry
Asked by kuldeepsingh33087 | 27 Jul, 2019, 11:35: AM
CBSE 11-science - Chemistry
Asked by vijaykumartecno348 | 06 Jul, 2019, 09:26: PM
CBSE 11-science - Chemistry
Asked by Taniyarana150 | 23 May, 2019, 12:01: AM







