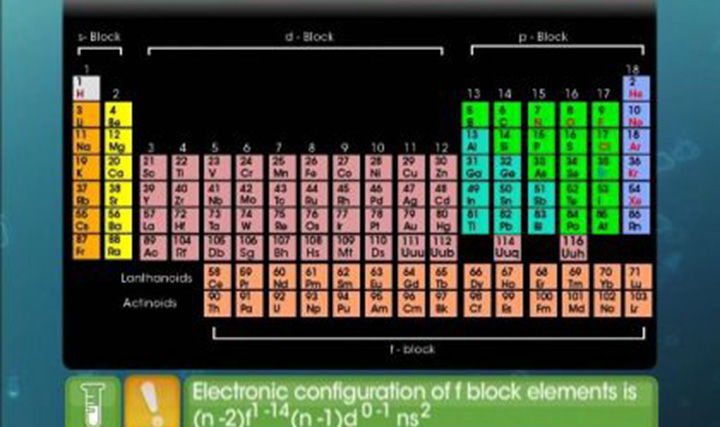
CBSE Class 12-science Chemistry Lanthanoids and Actinoids
TopperLearning’s CBSE Class 12 Science Chemistry The d-Block and f-Block Elements – Lanthanoids and Actinoids study resources enable you to study according to your convenience online. You can visit our online portal and access learning videos on the properties of lanthanoids and actinoids 24/7. Revise the electronic configuration of these d-block elements with our video explanations anytime.
Utilise our learning portal’s CBSE Class 12 Science Chemistry sample papers to revise different types of questions such as short answer questions and MCQs. In addition, learn online for your board exam easily with our textbook solutions, topic notes, practice question papers and more.
- Why the separation of actonoids are very difficult
- Why lanthanide show high oxidation state than actinide
- Why do Zr and Hf occur together in nature?
- Write down the most common oxidation state for lanthanoids.
- Write down the number of electrons in 4f, 5d and 6s orbital of Ce in +4 oxidation state.
- Write down the outer electronic configuration of Uranium.
- Why Ce4+ behaves as good oxidising agent although being a stable species?
- What is mischmetall and what is its use?
- Why it is very difficult to study the chemistry of Actinoid?
- Write the electronic configuration of the following elements of lanthanoid and their possible oxidation state other than +3: (a) Eu and (b) Yb.