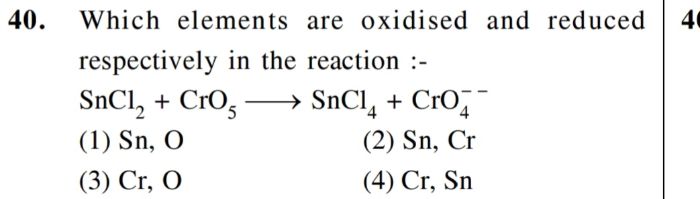CBSE Class 12-science Answered
Why d block elements forms complex compounds?
Asked by basib61203 | 08 Feb, 2024, 18:03: PM
Dear Student,
Because transition elements have a high charge density and small size of ions with a higher nuclear charge, they easily form complexes.
A complex is formed because there are open d-orbitals where a ligand can give up an electron.
Because of their strong polarizing ability, transition metal ions can draw lone pairs from other atoms to create complexes.
Answered by | 09 Feb, 2024, 09:45: AM
Concept Videos
CBSE 12-science - Chemistry
Asked by anubhutiupadhaya | 27 Feb, 2024, 16:28: PM
CBSE 12-science - Chemistry
Asked by basib61203 | 08 Feb, 2024, 18:03: PM
CBSE 12-science - Chemistry
Asked by ABHILASHA | 04 Mar, 2021, 02:26: AM
CBSE 12-science - Chemistry
Asked by ghoshmahadev037 | 20 Sep, 2020, 11:45: AM
CBSE 12-science - Chemistry
Asked by mufeedatvp2000 | 20 Apr, 2020, 14:53: PM
CBSE 12-science - Chemistry
Asked by patra04011965 | 25 Sep, 2019, 22:22: PM
CBSE 12-science - Chemistry
Asked by patra04011965 | 22 Sep, 2019, 13:42: PM
CBSE 12-science - Chemistry
Asked by jain.pradeep | 30 Aug, 2019, 08:09: AM
CBSE 12-science - Chemistry
Asked by Debdulal | 29 Aug, 2019, 16:46: PM
CBSE 12-science - Chemistry
Asked by Debdulal | 29 Aug, 2019, 16:44: PM