CBSE Class 11-science Answered
The enthalpy of combustion for H2, C (graphite) and CH4 are -285.8, -393.5, and -890.4 kJ/mol respectively. Calculate the standard enthalpy of formation ?Hf for CH4.
Asked by Topperlearning User | 15 Jun, 2016, 17:36: PM
We can write equation as,
C + 2H2 → CH4
C + 2H2 → CH4
Enthalpy of formation.
(1) H2(g) + 0.5 O2(g) → H2O(l) 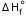 = -285.8 (kJ/mol)
= -285.8 (kJ/mol)
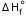 = -285.8 (kJ/mol)
= -285.8 (kJ/mol)(2) C(graphite) + O2(g) → CO2(g) 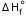 = -293.5 (kJ/mol)
= -293.5 (kJ/mol)
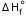 = -293.5 (kJ/mol)
= -293.5 (kJ/mol)Enthalpy of combustion methane
(3) CH4(g) + 2O2(g) →CO2(g) + 2H2O(l) 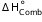 = -890.4(kJ/mol)
= -890.4(kJ/mol)
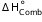 = -890.4(kJ/mol)
= -890.4(kJ/mol)Step1. Multiply equation 1 by 2
(1) H2(g) + 0.5 O2(g) → H2O(l) 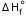 = -285.8 (kJ/mol) x 2
= -285.8 (kJ/mol) x 2
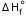 = -285.8 (kJ/mol) x 2
= -285.8 (kJ/mol) x 2(4) 2H2(g) + O2(g) → 2H2O(l) 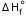 = -571.6 (kJ/mol)
= -571.6 (kJ/mol)
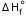 = -571.6 (kJ/mol)
= -571.6 (kJ/mol)Step2. To 4 add 2 and subtract 3
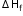 = 2 x (-285.8) + (-393.5) - (-890.4) = -74.7 (kJ/mol)
= 2 x (-285.8) + (-393.5) - (-890.4) = -74.7 (kJ/mol)
Answered by | 15 Jun, 2016, 19:36: PM
Concept Videos
CBSE 11-science - Chemistry
Asked by paulnaveed202 | 17 Dec, 2020, 12:36: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 13 Aug, 2014, 16:24: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 04 Jun, 2014, 13:23: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 14 Aug, 2014, 09:38: AM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 15 Jun, 2016, 17:36: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 14 Aug, 2014, 09:58: AM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 15 Jun, 2016, 17:35: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 14 Aug, 2014, 10:15: AM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 14 Aug, 2014, 10:41: AM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 15 Jun, 2016, 17:35: PM

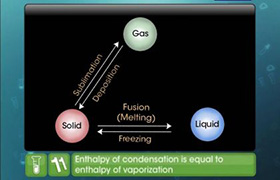


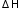 from: Enthalpy changes of formation and bond enthalpies.
from: Enthalpy changes of formation and bond enthalpies.