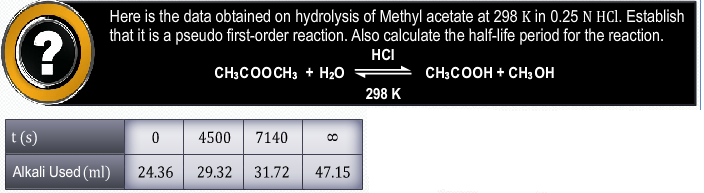
CBSE Class 12-science Answered
A reaction is 50% completed in 2 hours and 75% completed in 4 hours, What is the order of reaction ?
Asked by Deepak Singh | 04 May, 2015, 09:21: PM
First half reaction gets completed within 2 hours and next half reaction gets completed within next 2 hours. That means, t1/2 is indepedent of concentration. Hence, the order of reaction is first order reaction.
Answered by Prachi Sawant | 05 May, 2015, 08:41: AM
Concept Videos
CBSE 12-science - Chemistry
Asked by rchaitra1204 | 07 Sep, 2020, 09:43: AM
CBSE 12-science - Chemistry
Asked by arunhys123 | 04 Jul, 2020, 07:36: PM
CBSE 12-science - Chemistry
Asked by dhruvrana348 | 28 Jun, 2020, 08:58: AM
CBSE 12-science - Chemistry
Asked by lovemaan5500 | 06 Jan, 2020, 03:39: PM
CBSE 12-science - Chemistry
Asked by lovemaan5500 | 18 Sep, 2019, 10:02: PM
CBSE 12-science - Chemistry
Asked by sanjeet.kumar | 12 Mar, 2019, 02:20: PM
CBSE 12-science - Chemistry
Asked by manpreetkaur19971993 | 10 Jan, 2019, 07:06: AM
CBSE 12-science - Chemistry
Asked by rohitraman1115 | 22 Jul, 2018, 08:27: PM
CBSE 12-science - Chemistry
Asked by Topperlearning User | 27 Mar, 2014, 01:37: PM