CBSE Class 11-science Answered
give point of difference between specific heat and heat capacity ( as many as you can ) giving example of each
Asked by ppratim02 | 29 Jan, 2016, 08:31: PM
| Heat capacity | Specific heat capacity |
| The heat capacity of a body is the amount of heat energy required to raise its temperature by 1°C or 1 K. | The specific heat capacity of a substance is the amount of heat energy required to raise the temperature of unit mass of that substance through 1°C (or 1 K). |
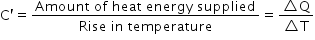 |
 Therefore,
 |
| It is denoted by symbol C′. | It is denoted by the symbol c. |
| It depends on the mass of the material. | It is independent of the mass of the material. |
| The SI unit of heat capacity is joule per kelvin (or J K−1). It is also written as joule per degree C (or J °C−1). | The SI unit of specific heat capacity is joule per kilogram per kelvin (or J kg−1K−1) or joule per kilogram per degree celsius (or J kg−1 °C−1). |
| Example: Heat capacity of water for 100 g is 4.179 J/°C | Example: Specific heat capacity of water is 4200 J kg-1 °C-1 = 4.2 J g-1 °C-1 |
Answered by Faiza Lambe | 30 Jan, 2016, 09:42: AM
Concept Videos
CBSE 11-science - Physics
Asked by arjunsah797 | 01 Mar, 2022, 03:01: PM
CBSE 11-science - Physics
Asked by sanjanapujar80 | 12 Jun, 2021, 10:29: AM
CBSE 11-science - Physics
Asked by shubhamgoyal1722 | 15 Dec, 2020, 11:18: AM
CBSE 11-science - Physics
Asked by Venkeylkm | 16 Jul, 2020, 04:12: PM
CBSE 11-science - Physics
Asked by pgskgokul | 06 Jul, 2020, 02:50: PM
CBSE 11-science - Physics
Asked by abhilashkulgude111 | 07 Jun, 2019, 09:05: PM
CBSE 11-science - Physics
Asked by Topperlearning User | 04 Jun, 2014, 01:23: PM
CBSE 11-science - Physics
Asked by Topperlearning User | 13 Apr, 2015, 03:28: PM
CBSE 11-science - Physics
Asked by Topperlearning User | 13 Apr, 2015, 03:19: PM
CBSE 11-science - Physics
Asked by Topperlearning User | 13 Apr, 2015, 03:20: PM




