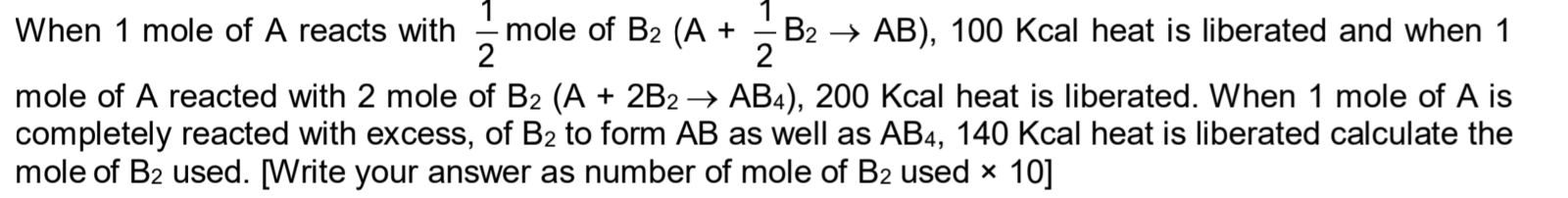CBSE Class 11-science Answered
Dissolving 120g of urea in 1000g of water gave a solution of density 1.12 g/ml . The molarity of solution is ?
Asked by arunavamitra50 | 05 May, 2018, 08:35: AM
Given:
Weight of urea= 120 g
weight of water= 1000 g
density= 1.2 g/ml
We know,
Density = Mass/Volume
Volume = Mass/ Density
= (120+1000)/ 1.12
= 1120/1.12
= 1000 ml
= 1 litre.
Molarity = No. of moles od solute/ Volume in litre
No. of moles = weight/ Miolecular weight
= 120/ 60
No. of moles = 2 mole
Molarity = 2/1
= 2 M
Answered by Varsha | 05 May, 2018, 06:22: PM
Concept Videos
CBSE 11-science - Chemistry
Asked by drhimasingh | 22 May, 2020, 11:39: AM
CBSE 11-science - Chemistry
Asked by nareshrajpurohit43109 | 22 May, 2020, 11:18: AM
CBSE 11-science - Chemistry
Asked by d6knx7qmw1 | 15 May, 2020, 10:37: PM
CBSE 11-science - Chemistry
Asked by sahadipa1975 | 02 May, 2020, 08:53: AM
CBSE 11-science - Chemistry
Asked by abhishek19362771 | 08 Apr, 2020, 03:48: PM
CBSE 11-science - Chemistry
Asked by anilsolanki2060 | 22 Feb, 2020, 10:12: AM
CBSE 11-science - Chemistry
Asked by pujakurmi22 | 11 Nov, 2019, 10:59: PM
CBSE 11-science - Chemistry
Asked by jkatwara | 14 Oct, 2019, 12:21: PM
CBSE 11-science - Chemistry
Asked by vikas.kochhar6 | 30 Aug, 2019, 03:58: PM
CBSE 11-science - Chemistry
Asked by pb_ckt | 19 May, 2019, 11:56: PM