CBSE Class 11-science - Formulation and Calculation Videos
Basic Concepts of Chemistry
This video contains practice questions and numerical problems based on the percentage composition and empirical formula.
More videos from this chapter
View All- empirical formula of glucose is
-
In this, if the options weren't there, couldn't it have been YX 2 (2 in subscript)

- 2g Feso4 is completely oxidised by 0.05M kmno4.what volume of kmno4 is required?
-
Please solve the qustion in the attachment
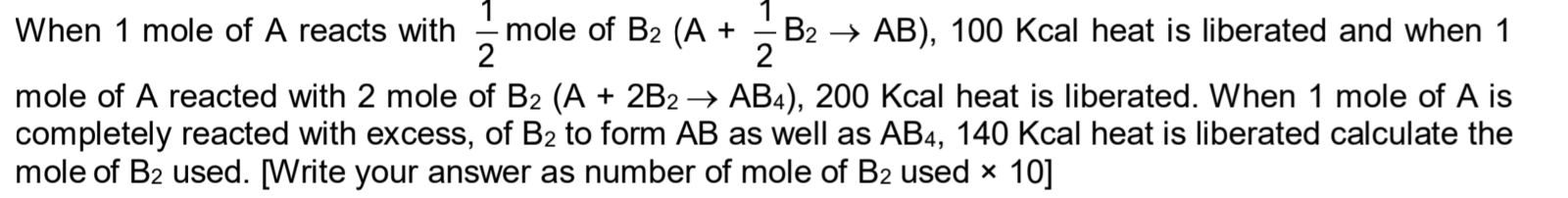
-
Sir Q.no 3

- what is the mole fraction of alcohol in 10% of alcohol w/w in water ?
-
ans with solutions

-
Q.no 3 =the relative abundance of various isotopes of silicon is as Si-28=92.23%, Si-29=4.67% & Si-30=3.10%. what is the average atomic mass of silicon.

- Vitamins C is essential fot prevention of scurvy combustion of 0.9g of vitamins C givez 0.2998g of Co2and 0.819g of H2OWhat is empirical formula of Vitamin C
- A compound of lead has the following percentage composition: Pb = 90.66%, O = 9.34%. Calculate the empirical formula of the compound.(Pb =207,O=16 )






