CBSE Class 11-science Answered
2 L of NH3 at 273K and 1 ATM pressure neutralised 134ml of a solution of H2SO4.What is the normality of the acid?
Asked by pb_ckt | 19 May, 2019, 23:56: PM
Given:
Volume; V = 1 litre
Pressure; P = 1 atm
Temparature; T = 273 K
n = ?
We know,
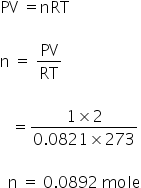
Meq of NH3 = Meq of H2SO4
0.0821× 1000= 134×Normality of an acid
Normality of an acid =
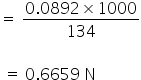
The normality of the acid is 0.6659 N.
Answered by Varsha | 20 May, 2019, 10:16: AM
Concept Videos
CBSE 11-science - Chemistry
Asked by rathodhamirbhai94 | 12 Jul, 2024, 21:34: PM
CBSE 11-science - Chemistry
Asked by drhimasingh | 22 May, 2020, 11:39: AM
CBSE 11-science - Chemistry
Asked by nareshrajpurohit43109 | 22 May, 2020, 11:18: AM
CBSE 11-science - Chemistry
Asked by d6knx7qmw1 | 15 May, 2020, 22:37: PM
CBSE 11-science - Chemistry
Asked by sahadipa1975 | 02 May, 2020, 08:53: AM
CBSE 11-science - Chemistry
Asked by abhishek19362771 | 08 Apr, 2020, 15:48: PM
CBSE 11-science - Chemistry
Asked by anilsolanki2060 | 22 Feb, 2020, 10:12: AM
CBSE 11-science - Chemistry
Asked by pujakurmi22 | 11 Nov, 2019, 22:59: PM
CBSE 11-science - Chemistry
Asked by jkatwara | 14 Oct, 2019, 12:21: PM
CBSE 11-science - Chemistry
Asked by vikas.kochhar6 | 30 Aug, 2019, 15:58: PM