CBSE Class 12-science Answered
why is glass considered as super cooled liquid....????
Asked by Potam Mahesh | 30 Jul, 2014, 10:19: PM
Glass has ability to flow as it is amorphous. It can be observed in the windows of old buildings. They are thicker at the bottom and thinner at the top. It shows that glass has the ability to flow which is normally showwn by liquids. Hence glass is reffered as super-cooled liquid.
Answered by Prachi Sawant | 31 Jul, 2014, 09:59: AM
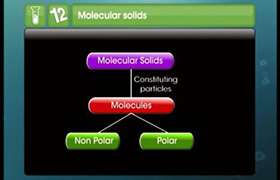
Concept Videos
CBSE 12-science - Chemistry
Asked by sardaarjii2012 | 31 Oct, 2021, 09:05: PM
CBSE 12-science - Chemistry
Asked by karennavarpriya | 20 Aug, 2021, 01:53: PM
CBSE 12-science - Chemistry
Asked by apoorvaks26 | 10 May, 2021, 09:24: AM
CBSE 12-science - Chemistry
Asked by someshmule5 | 05 Jan, 2021, 01:25: PM
CBSE 12-science - Chemistry
Asked by deepakatur454 | 16 Oct, 2020, 01:18: PM
CBSE 12-science - Chemistry
Asked by hemanthkumarbk2356 | 02 Aug, 2020, 12:16: PM
CBSE 12-science - Chemistry
Asked by harishshrivastava855 | 21 Jun, 2020, 09:26: PM
CBSE 12-science - Chemistry
Asked by manjurana04933 | 16 Apr, 2020, 11:00: AM
CBSE 12-science - Chemistry
Asked by Saransekar407 | 11 Mar, 2019, 06:47: PM
CBSE 12-science - Chemistry
Asked by yaag8432 | 05 Mar, 2019, 07:28: PM