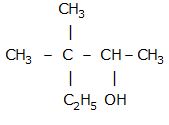CBSE Class 12-science Answered
why C-O bond in phenols is larger than that of alcohols.
Asked by pfffxy571 | 08 Jun, 2018, 10:27: AM
The question should be: why C-O bond in alcohols is larger than that of phenols.
Answer:
Phenol has a benzene ring with a -OH group attached to it, the lone pair of this oxygen of _OH group is in conjugation with the phenyl ring, hence the C-O bond will attain the partial double bond character, and the bond length of the double bond is shorter than the single bond.
Whereas in aliphatic alcohols there no such conjugation is there, hence the bond length of C-O of Alcohol is larger than that of the phenols.
Answered by Ramandeep | 08 Jun, 2018, 12:56: PM
Concept Videos
CBSE 12-science - Chemistry
Asked by guneetk44 | 28 Apr, 2024, 10:39: AM
CBSE 12-science - Chemistry
Asked by akhilmaigur987 | 20 May, 2020, 14:55: PM
CBSE 12-science - Chemistry
Asked by aayushkumargupta3 | 10 Apr, 2020, 22:56: PM
CBSE 12-science - Chemistry
Asked by chauhanparth974 | 08 Mar, 2020, 09:34: AM
CBSE 12-science - Chemistry
Asked by ajaysankhala051 | 03 Mar, 2020, 23:33: PM
CBSE 12-science - Chemistry
Asked by pfffxy571 | 08 Jun, 2018, 10:27: AM
CBSE 12-science - Chemistry
Asked by Girijeshpandey.rjil | 23 Apr, 2018, 19:16: PM
CBSE 12-science - Chemistry
Asked by hbohra67 | 04 Apr, 2018, 03:53: AM
CBSE 12-science - Chemistry
Asked by Balbir | 03 Apr, 2018, 20:26: PM
CBSE 12-science - Chemistry
Asked by raul.chintakindi14 | 14 Mar, 2018, 11:58: AM