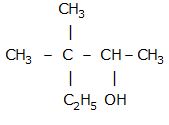CBSE Class 12-science Answered
Introduction of alcohols
Asked by akhilmaigur987 | 20 May, 2020, 14:55: PM
Alcohols:
Alcohols are compounds formed when a hydrogen atom in a hydrocarbon is replaced by the –OH group. An alcohol contains one or more hydroxyl (-OH) group(s) directly attached to carbon atom(s) of an aliphatic system.
Alcohol may be classified as mono-, di-, tri-or polyhydric compounds depending on whether they contain one, two, three or many hydroxyl groups,respectively,in their structures.

According to IUPAC, alcohols get their name from the alkanes from which they are derived by substituting the ‘e’ of the alkane with the suffix ‘–ol’.
The position of the substituents is indicated by numbers. Theparent chain is numbered from the end nearest to the –OH group.
The positions of the –OH group and other substituents are denoted using the numbers of carbon atoms to which they are attached.
The prefix di,tri, etc. is usedto indicate the number of –OH groups.

Answered by Ramandeep | 20 May, 2020, 19:15: PM
Concept Videos
CBSE 12-science - Chemistry
Asked by guneetk44 | 28 Apr, 2024, 10:39: AM
CBSE 12-science - Chemistry
Asked by akhilmaigur987 | 20 May, 2020, 14:55: PM
CBSE 12-science - Chemistry
Asked by aayushkumargupta3 | 10 Apr, 2020, 22:56: PM
CBSE 12-science - Chemistry
Asked by chauhanparth974 | 08 Mar, 2020, 09:34: AM
CBSE 12-science - Chemistry
Asked by ajaysankhala051 | 03 Mar, 2020, 23:33: PM
CBSE 12-science - Chemistry
Asked by pfffxy571 | 08 Jun, 2018, 10:27: AM
CBSE 12-science - Chemistry
Asked by Girijeshpandey.rjil | 23 Apr, 2018, 19:16: PM
CBSE 12-science - Chemistry
Asked by hbohra67 | 04 Apr, 2018, 03:53: AM
CBSE 12-science - Chemistry
Asked by Balbir | 03 Apr, 2018, 20:26: PM
CBSE 12-science - Chemistry
Asked by raul.chintakindi14 | 14 Mar, 2018, 11:58: AM