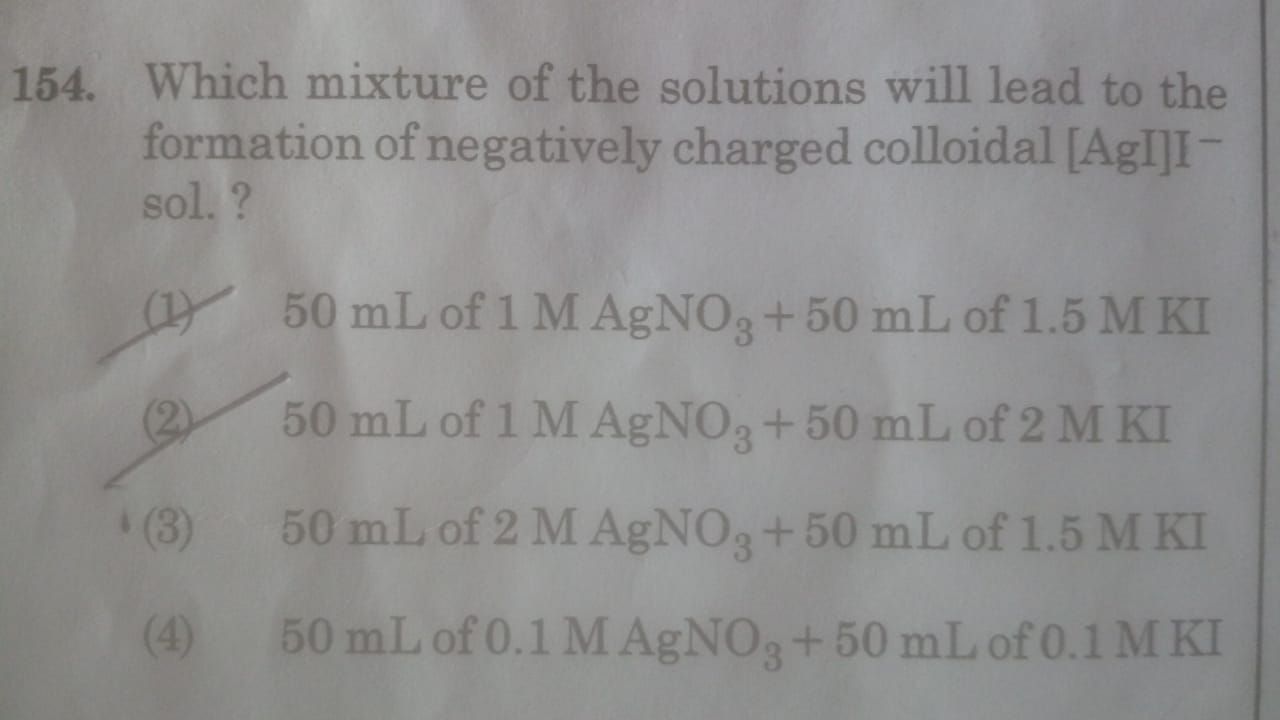CBSE Class 12-science Answered
what is hardy-schulze rule?
Asked by Ayush kumar | 09 Mar, 2014, 10:52: PM
The quantity of the electrolyte which is required to coagulate a definite amount of a colloidal solution depends upon the valency of the coagulating ion(ion having a charge opposite to that of the colloidal particles). This observation of Hardy and Schulze is known as Hardy Schulze Law, the main points of which may be stated as follows:
(i) The effective ions of the electroyte in bringing about coagulation are thise which carry charge opposite to that of the colloidal particles. These ions are called coagulating ions or flocculating ions.
(ii) Greater is the valency of the coagulating or the flocculating ion, greater is its power to bring about coagulation.
Answered by | 10 Mar, 2014, 11:06: AM
Application Videos
Concept Videos
CBSE 12-science - Chemistry
Asked by mallavaramchandrika6 | 29 Sep, 2021, 05:27: PM
CBSE 12-science - Chemistry
Asked by Akshij Nanda | 11 Mar, 2021, 10:21: AM
CBSE 12-science - Chemistry
Asked by sivakapuganti1 | 26 Aug, 2020, 08:57: PM
CBSE 12-science - Chemistry
Asked by spoorthysaienelluri | 23 May, 2020, 11:07: AM
CBSE 12-science - Chemistry
Asked by shreemuvijihari | 18 May, 2020, 06:29: PM
CBSE 12-science - Chemistry
Asked by vermahitesh124 | 12 May, 2020, 07:58: AM
CBSE 12-science - Chemistry
Asked by yogeshsulakh | 07 Feb, 2020, 09:28: AM
CBSE 12-science - Chemistry
Asked by tlb2bpartner | 21 Aug, 2019, 10:28: PM
CBSE 12-science - Chemistry
Asked by ntg432000 | 22 May, 2019, 08:10: PM
CBSE 12-science - Chemistry
Asked by kripanjalihimansu | 01 Mar, 2019, 04:16: PM