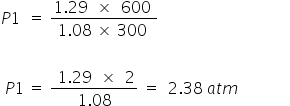CBSE Class 11-science Answered

N2O4(g) 2NO2(g)
At start 100/92 = 1.08mol 0
At equilibrium 80/92 = 0.86 20/46 mol= 0.43 mol
According to ideal gas equation, at two condition
At 300K P0V = n0RT0
1 × V = 1.08 × R × 300 -------------- (i)
At 600K P1V = n1RT1
P1 × V = (0.86 + 0.43) × R ×600 ------------(ii)
Divide (ii) by (i)