CBSE Class 11-science Answered
For a reaction, A + B → 2C
2 moles of A and 3 moles of B are allowed to react. If equilibrium constant is 4 at 400° C,
then the mole of C at equilibrium is
Asked by tanuj2808 | 25 Sep, 2019, 10:18: AM
Let's suppose volume= 1 L
In this case, Number of moles=Concentration
State of equilibrium can be expressed as-
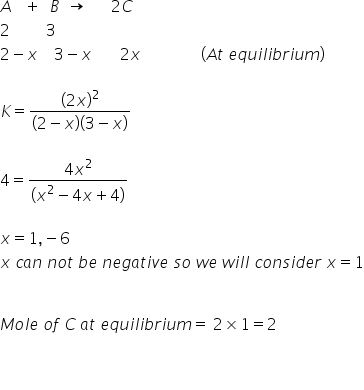
Answered by Ravi | 25 Sep, 2019, 18:14: PM
Concept Videos
CBSE 11-science - Chemistry
Asked by gouravvv641 | 16 Aug, 2022, 21:25: PM
CBSE 11-science - Chemistry
Asked by mangalchandrj79 | 21 May, 2022, 16:38: PM
CBSE 11-science - Chemistry
Asked by veenatripathi | 28 May, 2020, 09:03: AM
CBSE 11-science - Chemistry
Asked by ABHILASHA | 24 Oct, 2019, 06:27: AM
CBSE 11-science - Chemistry
Asked by tanuj2808 | 25 Sep, 2019, 10:18: AM
CBSE 11-science - Chemistry
Asked by jhajuhi19 | 02 Jun, 2019, 23:55: PM
CBSE 11-science - Chemistry
Asked by ntg432000 | 26 Apr, 2019, 16:31: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 24 Apr, 2015, 13:55: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 24 Apr, 2015, 14:07: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 24 Apr, 2015, 14:31: PM









