NEET Class neet Answered

(a) ( ΔS)isothermal > ( ΔS )adiabatic
Above statement is true .
Change in entropy of adiabatic is zero.
Change in entropy for isothermal expansion process is greater than zero becuase heat is absorbed by system .
Hene change in entropy of isothermal process is greater than that of adiabatic process .
----------------------------------------
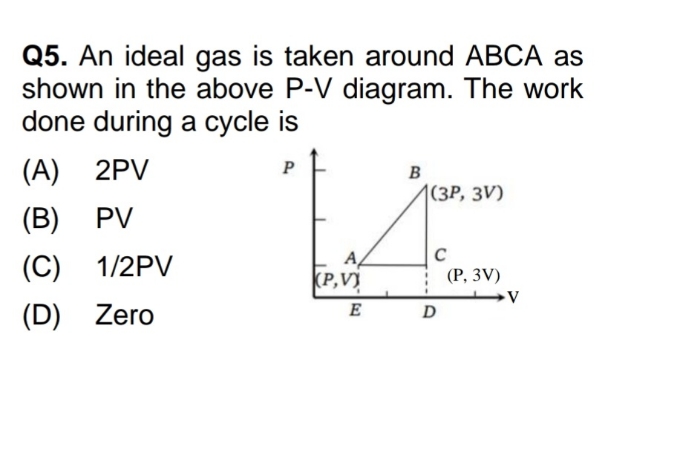
(b) TA = TB
It is true because process AB is isothermal process
--------------------------------------
(c) Wisothermal > Wadiabatic
It is true because workdone by isothermal process is performed by absorbing external heat energy whereas in adiabatic process work is done by utilizing internal energy..
-------------------------------------------
(d) TC > TA
In adiabatic process AC , gas expands using the internal energy and internal energy decreases when gas expands .
Hence TC < TA