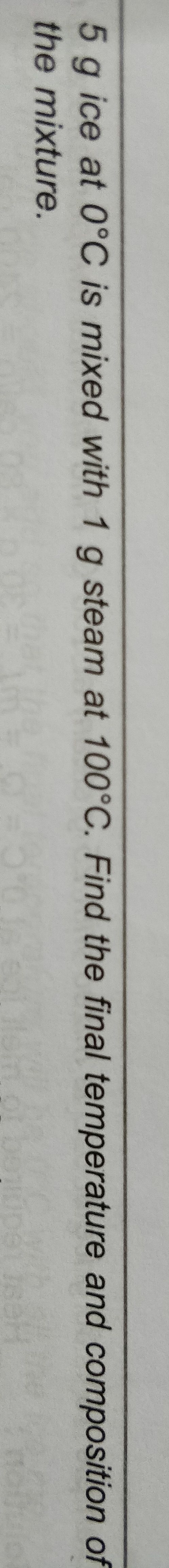NEET Class neet Answered
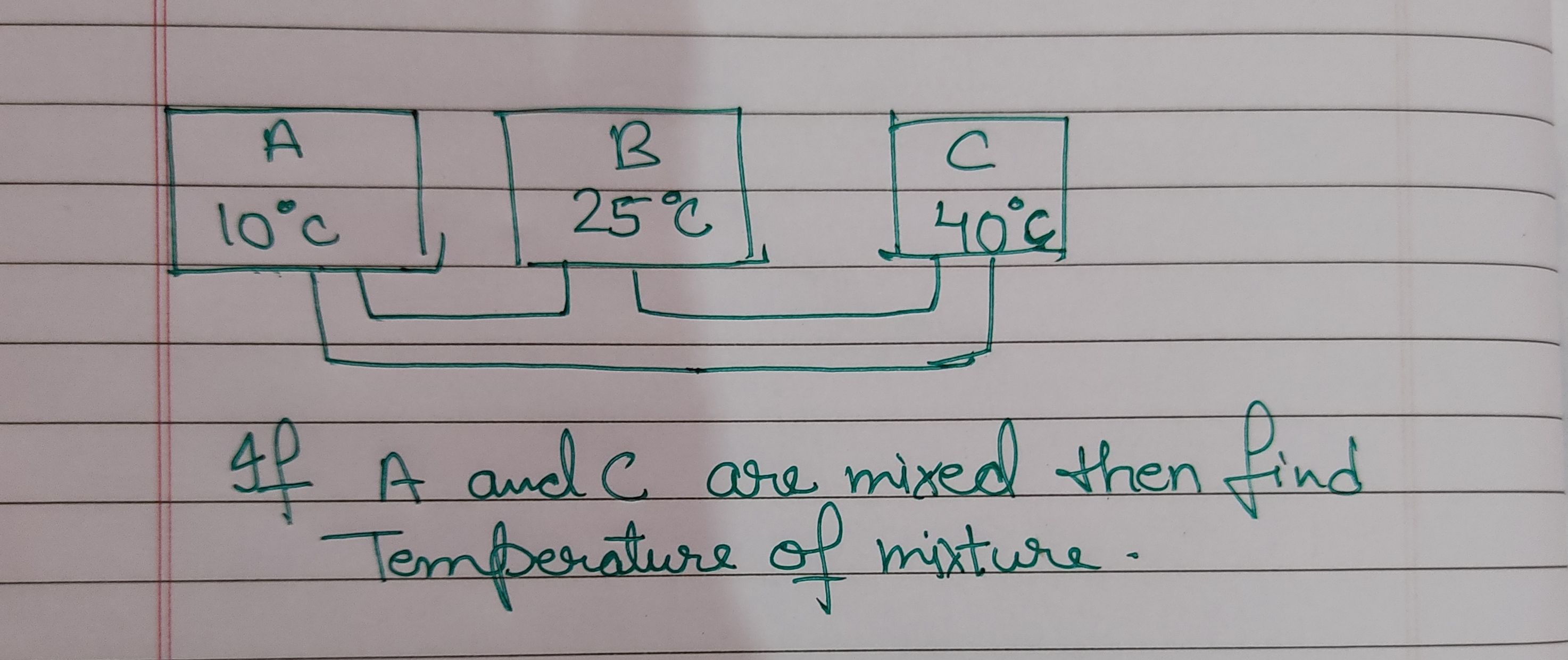
If liquid in A and C are mixed, find temperature of mixture.
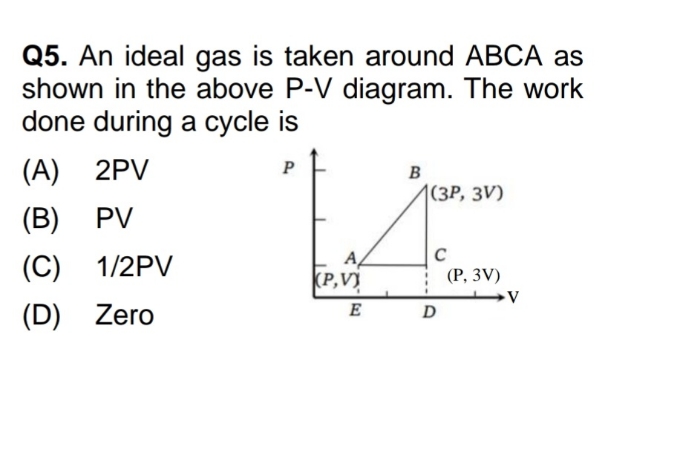
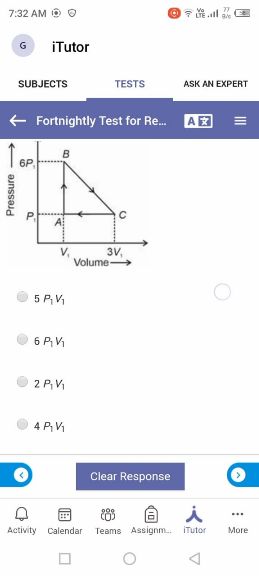
(Thermodynamics Chapter)
Asked by jhajuhi19 | 18 Aug, 2021, 03:21: AM
Let T be be the final equilibrium temperature after mixing liquid-A and liquid-C
Heat energy gained by liquid-A = mA Cp ( T - 10 )
Heat energy lost by liquid-C = mC Cp ( 40 - T )
By conservation of energy Heat gain equals heat loss.
If we assume masses of liquids are equal and Specif heats are equal ( A and C are same liquid) , then by conservation of energy
mA Cp ( T - 10 ) = mC Cp ( 40 - T )
(T-10) = ( 40 - T )
from above expression we get , T = 25o C
Answered by Thiyagarajan K | 18 Aug, 2021, 08:36: AM
NEET neet - Physics
Asked by yadavaradhana9335 | 19 Feb, 2024, 16:53: PM
NEET neet - Physics
Asked by sujitjana971 | 18 Dec, 2022, 17:23: PM
NEET neet - Physics
Asked by takshitashu46 | 09 Feb, 2022, 18:01: PM
NEET neet - Physics
Asked by jhajuhi19 | 18 Aug, 2021, 03:21: AM
NEET neet - Physics
Asked by 22bakugoku | 21 Apr, 2019, 17:23: PM
NEET neet - Physics
Asked by 22bakugoku | 21 Apr, 2019, 17:23: PM