CBSE Class 10 Answered
mam/sir, I made one formula to find the number of covalent bonds between the hydrocarbon so please will you tell me that it really works or not

Asked by krishdabhoya2003 | 21 Feb, 2019, 17:52: PM
Yes, but it would work only to find the number of sigma bonds in a hydrocarbons not pi bonds.
Covalent bond has two types (1) sigma bond formed by head-on overlapping hence it is strong bond (2) pi-bond formed by lateral overlapping, hence it is weak than sigma bond.
(1) In the case of alkane,
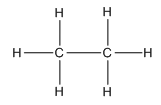
C2H6
Cn-1Hn = (2-1) +6 = 7 sigma covalent bonds.
(2) In the case of alkene,
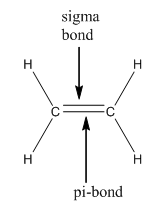
C2H4
Cn-1Hn = (2-1) +4 = 5 sigma covalent bonds.
This formula gives the only number of sigma bonds, not pi-bonds
Best way to find out,
The number of covalent bonds in alkane = Cn-1Hn
C2H6
Cn-1Hn = (2-1) +6 = 7
The number of covalent bonds in alkene = (Cn-1Hn) +1
C2H4
[Cn-1Hn]+1
= [(2-1) +4] + 1 = 5 + 1 = 6
The number of covalent bonds in alkyne = (Cn-1Hn) +2
C2H2
[Cn-1Hn ]+2= [(2-1) +2] +2 = 3+ 2 = 5
Answered by Ramandeep | 22 Feb, 2019, 11:50: AM
Application Videos
Concept Videos
CBSE 10 - Chemistry
Asked by psaisruthi10012009 | 07 Jun, 2024, 11:09: AM
CBSE 10 - Chemistry
Asked by tialempongen177 | 09 Sep, 2020, 23:44: PM
CBSE 10 - Chemistry
Asked by seeni2005 | 05 Jul, 2020, 21:53: PM
CBSE 10 - Chemistry
Asked by subbukum | 04 Feb, 2020, 11:46: AM
CBSE 10 - Chemistry
Asked by navjotsinghdadwal | 01 Dec, 2019, 21:57: PM
CBSE 10 - Chemistry
Asked by 9886761796hmh | 17 Oct, 2019, 20:10: PM
CBSE 10 - Chemistry
Asked by ashishaman25082004 | 15 Sep, 2019, 22:01: PM
CBSE 10 - Chemistry
Asked by dr.sudhiguptajdmd74 | 23 Jul, 2019, 09:18: AM
CBSE 10 - Chemistry
Asked by labheshvaidya | 19 Apr, 2019, 15:38: PM
CBSE 10 - Chemistry
Asked by krishdabhoya2003 | 21 Feb, 2019, 17:52: PM









