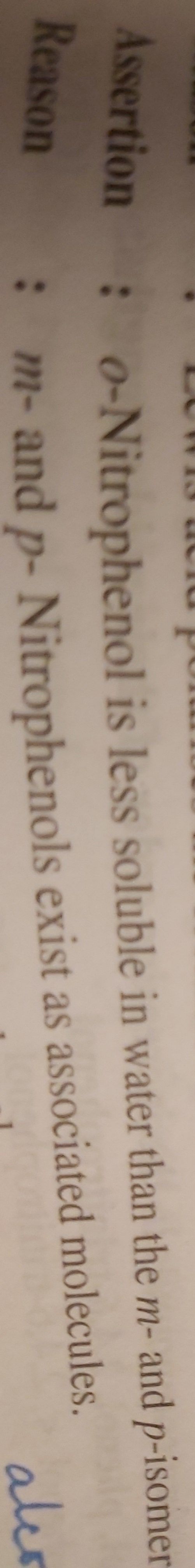CBSE Class 12-science Answered
How phenol is used in the manufacture of drugs like aspirin?
Asked by Topperlearning User | 27 Mar, 2014, 15:43: PM
- Phenol first needs to be treated with NaOH that will give sodium phenoxide.
- Sodium phenoxide when treated with CO2 at 400K under 4-7 atm pressure followed by acidification gives 2-hydroxy benzoic acid (salicylic acid as the main product).
- Salicyclic acid is the starting material for the manufacture of 2-Acetoxybenzoic acid(aspirin) which is used for relieving pain and to bring down the body temperature.
Answered by | 27 Mar, 2014, 17:43: PM
Concept Videos
CBSE 12-science - Chemistry
Asked by kavitabawane190 | 08 Mar, 2024, 17:24: PM
CBSE 12-science - Chemistry
Asked by rp0055293 | 07 Feb, 2024, 08:28: AM
CBSE 12-science - Chemistry
Asked by vipulverma | 14 Feb, 2022, 16:44: PM
CBSE 12-science - Chemistry
Asked by kaziryan.05 | 23 Jun, 2021, 20:02: PM
CBSE 12-science - Chemistry
Asked by Rg598555 | 30 Oct, 2019, 22:35: PM
CBSE 12-science - Chemistry
Asked by jain.pradeep | 30 Aug, 2019, 16:13: PM
CBSE 12-science - Chemistry
Asked by Topperlearning User | 27 Mar, 2014, 15:43: PM
CBSE 12-science - Chemistry
Asked by Topperlearning User | 07 Jun, 2016, 15:12: PM
CBSE 12-science - Chemistry
Asked by Topperlearning User | 07 Jun, 2016, 15:17: PM
CBSE 12-science - Chemistry
Asked by Topperlearning User | 27 Mar, 2014, 16:54: PM