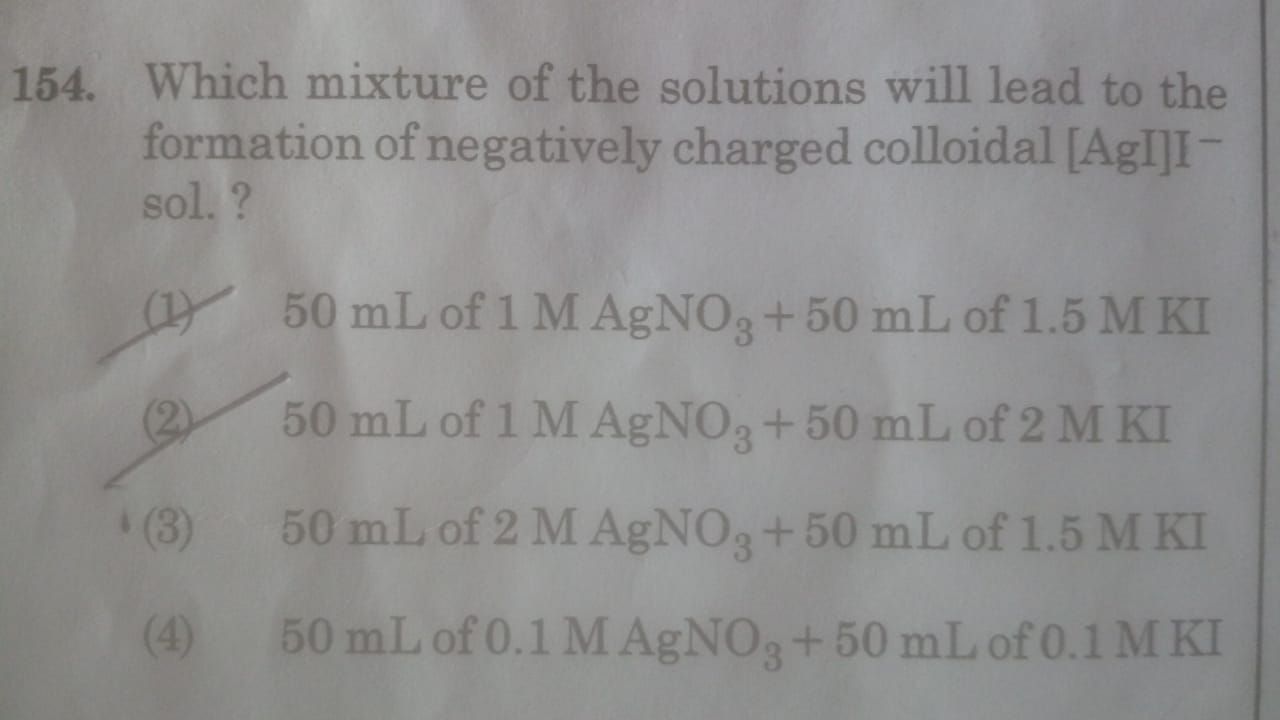CBSE Class 12-science Answered
Absorption generally refers to two phenomena which are largely unrelated. In one case, it refers to when atoms, molecules, or ions enter some bulk phase - gas, liquid or solid material. For instance, a sponge absorbs water when it is dry.Adsorption is similar, but refers to a surface rather than a volume: adsorption is a process that occurs when a gas or liquid solute accumulates on the surface of a solid or, more rarely, a liquid (adsorbent), forming a molecular or atomic film (the adsorbate). It is different from absorption, in which a substance diffuses into a liquid or solid to form a solution.
Similar to surface tension, adsorption is a consequence of surface energy. In a bulk material, all the bonding requirements (be they ionic, covalent, or metallic) of the constituent atoms of the material are filled by other atoms in the material. However, atoms on the surface of the adsorbent are not wholly surrounded by other adsorbent atoms and therefore can attract adsorbates. The exact nature of the bonding depends on the details of the species involved, but the adsorption process is generally classified as physisorption (characteristic of weak van der Waals forces) or chemisorption (characteristic of covalent bonding).