CBSE Class 11-science Answered
The bomb calorimeter consists primarily of the sample, oxygen, the stainless steel bomb, and water.
The dewar prevents heat flow from the calorimeter to the rest of the universe, i.e.,
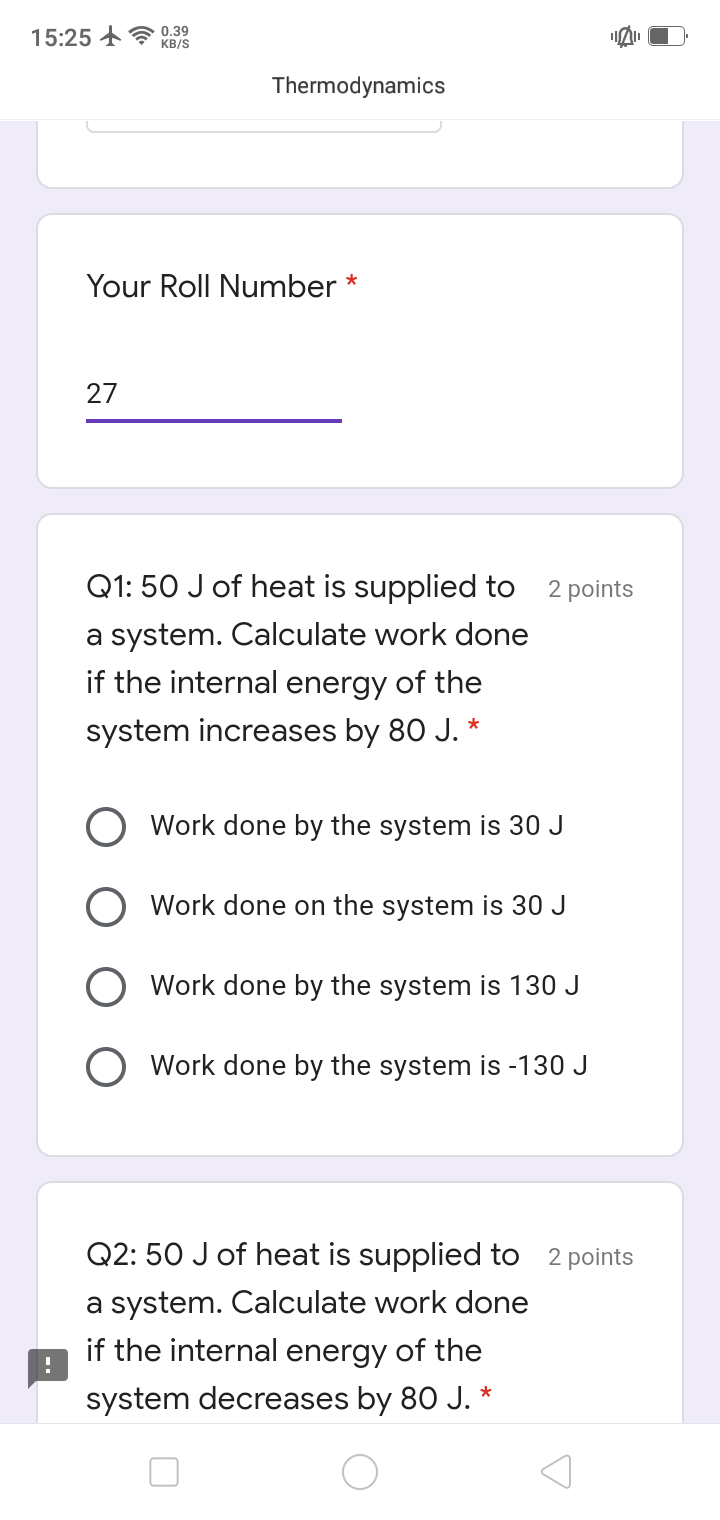
qcalorimeter = 0
Since the bomb is made from stainless steel, the combustion reaction occurs at constant volume and there is no work, i.e.,
wcalorimeter = -p dV = 0
Thus, the change in internal energy, ∆U, for the calorimeter is zero
∆Ucalorimeter = qcalorimeter + wcalorimeter = 0
The thermodynamic interpretation of this equation is that the calorimeter is isolated from the rest of the universe.
Since the calorimeter is isolated from the rest of the universe, we can define the reactants (sample and oxygen) to be the system and the rest of the calorimeter (bomb and water) to be the surroundings. The change in internal energy of the reactants upon combustion can be calculated from
Since the process if constant volume, dV = 0. Thus, recognizing the definition of heat capacity Cv yields
dUsys = – Cv dT
Assuming Cv to be independent of T over small temperature ranges, this expression can be integrated to give
∆U = – Cv dT
where Cv is the heat capacity of the surroundings, i.e., the water and the bomb.
From above expression we can calculate change in internal energy.