CBSE Class 12-science Answered
anthinodes lanthinodes
Asked by onkaronkar618 | 12 Oct, 2020, 23:38: PM
All lanthanides are f-block elements, corresponding to the filling of the 4f electron shell. The actinide or actinoid series encompasses the 15 chemical elements that lie between actinium and lawrencium included on the periodic table, with atomic numbers 89 - 103.
Answered by Ramandeep | 15 Oct, 2020, 16:04: PM
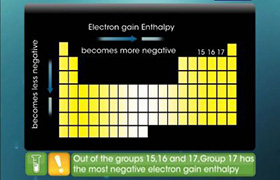
Concept Videos
CBSE 12-science - Chemistry
Asked by skumkum976 | 08 May, 2021, 15:49: PM
CBSE 12-science - Chemistry
Asked by manivannanbalakrishnan52 | 09 Dec, 2020, 22:06: PM
CBSE 12-science - Chemistry
Asked by onkaronkar618 | 12 Oct, 2020, 23:38: PM
CBSE 12-science - Chemistry
Asked by contactus.topperlearning | 13 Sep, 2020, 13:21: PM
CBSE 12-science - Chemistry
Asked by Daisysnaitz | 24 Apr, 2020, 01:07: AM
CBSE 12-science - Chemistry
Asked by minipkda | 22 May, 2018, 06:42: AM
CBSE 12-science - Chemistry
Asked by Topperlearning User | 09 Jun, 2014, 16:03: PM
CBSE 12-science - Chemistry
Asked by Topperlearning User | 07 Jun, 2016, 11:05: AM
CBSE 12-science - Chemistry
Asked by Topperlearning User | 09 Jun, 2014, 16:38: PM
CBSE 12-science - Chemistry
Asked by Topperlearning User | 10 Jun, 2014, 09:24: AM