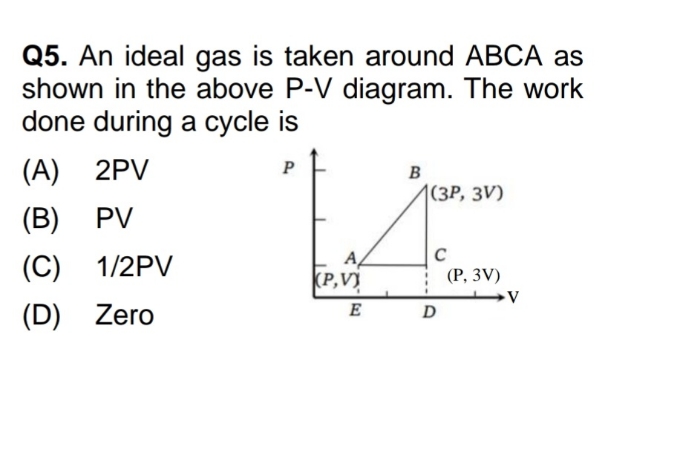
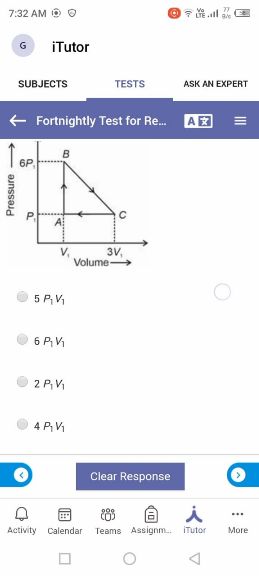
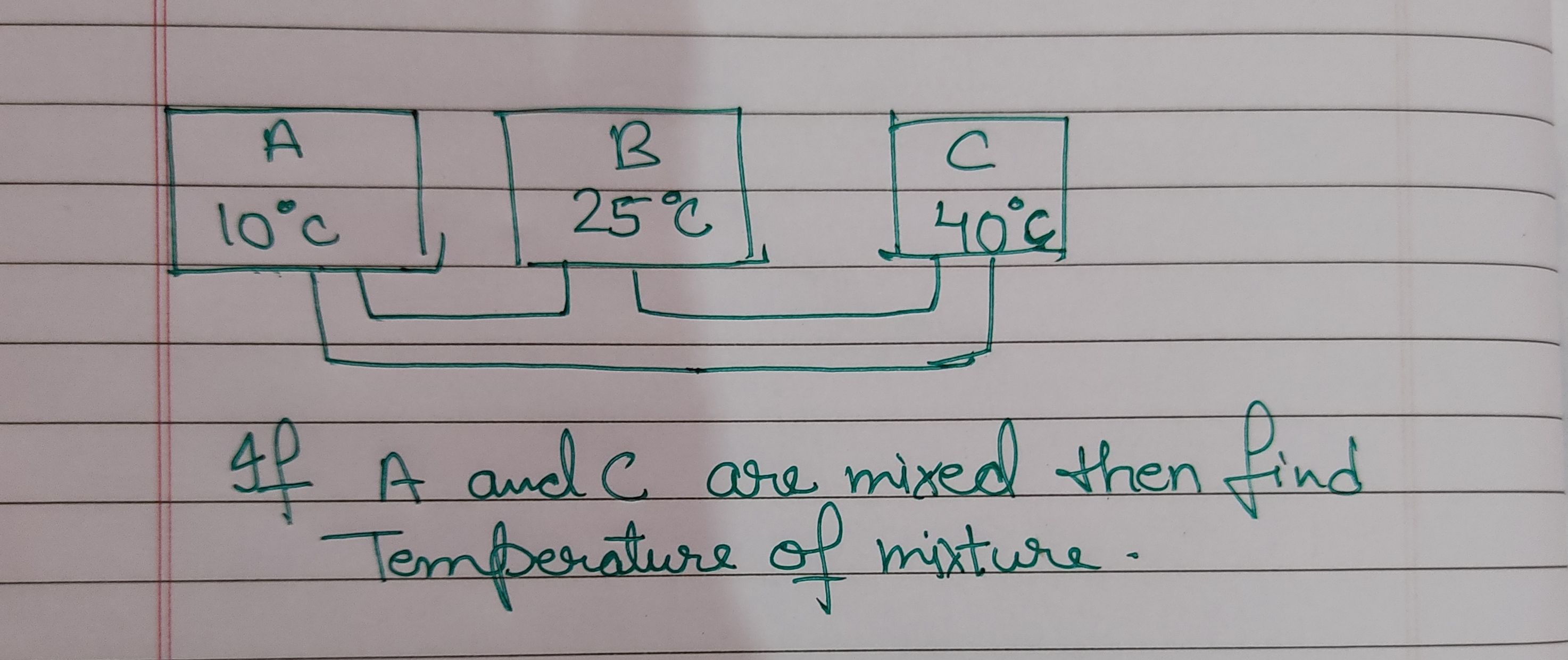
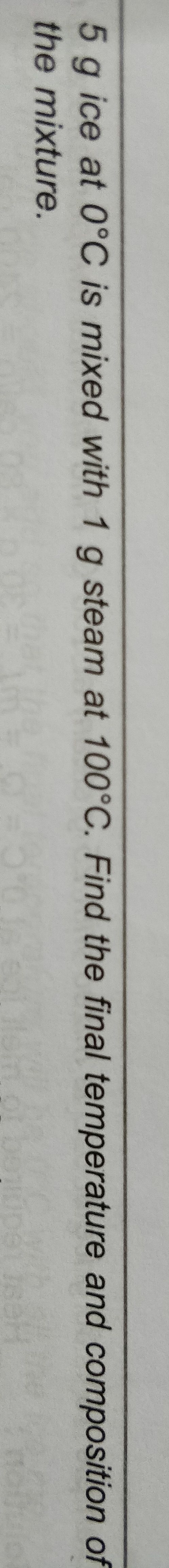NEET Class neet Answered
A container is filled with 20 moles of an ideal gas at absolute temperature T. When heat is supplied to gas temperature remains constant but 8 moles dissociate into atoms. Heat energy given to gas is-(1)4RT, (2)6RT, (3)3RT, (4)5RT
Asked by patra04011965 | 16 Mar, 2019, 12:20: PM
A container filled with n1 = 20 moles of ideal diatomic gas at absolute temperature T. when heat is supplied to gas temperature remains constant but n2 = 8 molesdissociated into atoms. Heat energy given to gas is Solution Since the gas is enclosed in a vessel, therefore, during heating process, volume of the gas remains constant. Hence, no work is done by the gas. It means heat supplied to the gas is used to increase its internal energy only. Initial internal energy of the gas is U1 =n1(5/2 RT ). Since n moles get dissociated into atoms, therefore, after heating, vessel contains (n1 − n2) moles of diatomic gas and 2n2 moles of a mono-atomic gas. Hence the internal energy for the gas, after heating, will be equal to U2 =(n1 − n2)5/2RT +2n2*3/2RT = 5/2n1RT + n2RT/2
Hence, the heat supplied is equal to the increase in internal energy
U2-U1 = 4RT
Answered by Ankit K | 16 Mar, 2019, 19:47: PM
NEET neet - Physics
Asked by yadavaradhana9335 | 19 Feb, 2024, 16:53: PM
NEET neet - Physics
Asked by sujitjana971 | 18 Dec, 2022, 17:23: PM
NEET neet - Physics
Asked by takshitashu46 | 09 Feb, 2022, 18:01: PM
NEET neet - Physics
Asked by jhajuhi19 | 18 Aug, 2021, 03:21: AM
NEET neet - Physics
Asked by 22bakugoku | 21 Apr, 2019, 17:23: PM
NEET neet - Physics
Asked by 22bakugoku | 21 Apr, 2019, 17:23: PM