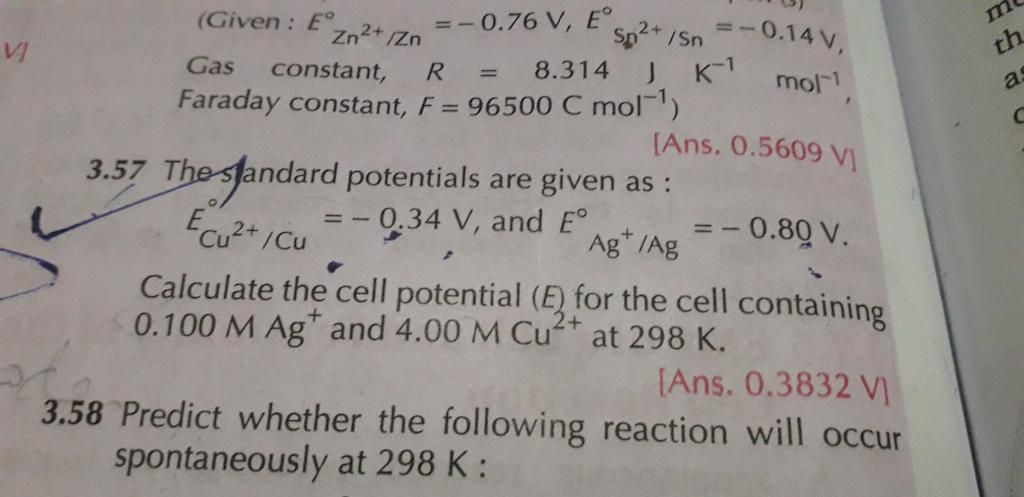CBSE Class 12-science Answered
3.52 numerical plz
Asked by lovemaan5500 | 19 Aug, 2019, 23:12: PM
Given:
ECu2+/Cu = −0.337 V
EMg2+/Mg = −2.37 V
F = 96500 C/mol
Net reaction,
Mg(s) + Cu2+(aq) → Mg2+(aq) + Cu(s)
E0cell = −0.337 − 2.37
= 2.74 V
Nernst equation;
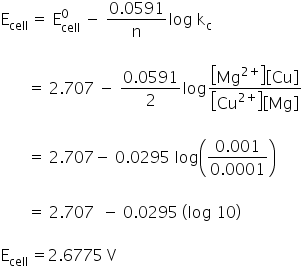
emf of the cell is 2.6775 V
Answered by Varsha | 20 Aug, 2019, 12:45: PM
Concept Videos
CBSE 12-science - Chemistry
Asked by routraypriyanka255 | 04 Jun, 2024, 23:43: PM
CBSE 12-science - Chemistry
Asked by avaneesh5116 | 07 Aug, 2020, 17:24: PM
CBSE 12-science - Chemistry
Asked by harshpareek696 | 01 Aug, 2020, 16:01: PM
CBSE 12-science - Chemistry
Asked by sulaikhasulu393 | 22 Jun, 2020, 08:52: AM
CBSE 12-science - Chemistry
Asked by vasudesetti123 | 23 May, 2020, 20:13: PM
CBSE 12-science - Chemistry
Asked by sakthisivasakthi1978 | 31 Oct, 2019, 21:53: PM
CBSE 12-science - Chemistry
Asked by lovemaan5500 | 19 Aug, 2019, 23:14: PM
CBSE 12-science - Chemistry
Asked by lovemaan5500 | 19 Aug, 2019, 23:13: PM
CBSE 12-science - Chemistry
Asked by lovemaan5500 | 19 Aug, 2019, 23:12: PM
CBSE 12-science - Chemistry
Asked by lovemaan5500 | 19 Aug, 2019, 23:11: PM