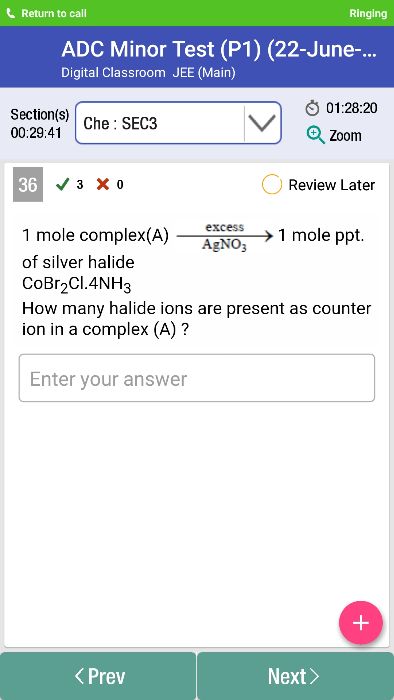
CBSE Class 12-science Answered
What is outer orbital complex and inner orbital complex ?
Please explain this with example
Asked by ADITYASEN704 | 11 Jun, 2016, 11:30: PM
The coordination number of central metal in these complexes is 6 having d2sp3 hybridisation. The complexes formed, if have inner d orbitals are called low spin complexes or inner orbital complexes and if having outer d orbitals are called high spin or outer orbital complex. The inner d orbitals are diamagnetic or less paramagnetic in nature hence, they are called low spin complexes.
Example:
Low spin complex: [Fe(CN)6]3-
High spin complex: [CoF3]3
Answered by Vaibhav Chavan | 12 Jun, 2016, 12:00: AM
Concept Videos
CBSE 12-science - Chemistry
Asked by prathyushagn1 | 09 Dec, 2020, 08:12: AM
CBSE 12-science - Chemistry
Asked by ABHILASHA | 31 Aug, 2020, 08:24: PM
CBSE 12-science - Chemistry
Asked by sha.bijoy17 | 07 Aug, 2020, 11:55: AM
CBSE 12-science - Chemistry
Asked by Shambhuhd79 | 22 Jun, 2020, 11:09: AM
CBSE 12-science - Chemistry
Asked by jain.pradeep | 19 Feb, 2020, 09:20: AM
CBSE 12-science - Chemistry
Asked by smit230503 | 04 Feb, 2020, 08:56: PM
CBSE 12-science - Chemistry
Asked by monishadubey202 | 08 Jan, 2020, 03:42: PM
CBSE 12-science - Chemistry
Asked by Chakshu29saini | 17 Sep, 2019, 06:19: PM
CBSE 12-science - Chemistry
Asked by bjayanta | 24 Mar, 2019, 08:56: PM
CBSE 12-science - Chemistry
Asked by himanshuneb | 28 Jan, 2019, 10:33: PM