CBSE Class 12-science Answered
Solve this
Asked by Shambhuhd79 | 22 Jun, 2020, 11:09: AM
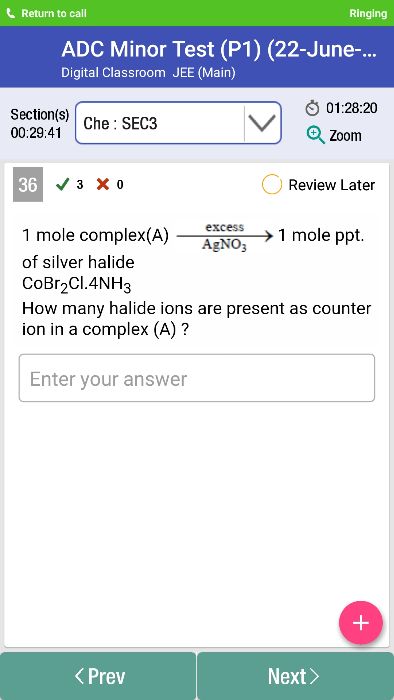
Coordination compound is [CoBr2(NH3)4] Cl
AgNO3 reacts with this complex and forms AgCl and [CoBr2(NH3)4] NO3
Counter ion is the ion which does not take part in coordination number of complex and balances the charge of cooordination sphere. Here Cl is the ion that is outside coordinationn sphere and not taking part in coordination number. So, one halide ion is counter ion in 1 complex compound.
Answered by Ravi | 22 Jun, 2020, 18:12: PM
Concept Videos
CBSE 12-science - Chemistry
Asked by prathyushagn1 | 09 Dec, 2020, 08:12: AM
CBSE 12-science - Chemistry
Asked by ABHILASHA | 31 Aug, 2020, 20:24: PM
CBSE 12-science - Chemistry
Asked by sha.bijoy17 | 07 Aug, 2020, 11:55: AM
CBSE 12-science - Chemistry
Asked by Shambhuhd79 | 22 Jun, 2020, 11:09: AM
CBSE 12-science - Chemistry
Asked by jain.pradeep | 19 Feb, 2020, 09:20: AM
CBSE 12-science - Chemistry
Asked by smit230503 | 04 Feb, 2020, 20:56: PM
CBSE 12-science - Chemistry
Asked by monishadubey202 | 08 Jan, 2020, 15:42: PM
CBSE 12-science - Chemistry
Asked by Chakshu29saini | 17 Sep, 2019, 18:19: PM
CBSE 12-science - Chemistry
Asked by bjayanta | 24 Mar, 2019, 20:56: PM
CBSE 12-science - Chemistry
Asked by himanshuneb | 28 Jan, 2019, 22:33: PM