CBSE Class 12-science Answered
Pl ans

Asked by jain.pradeep | 19 Feb, 2020, 09:20: AM
The correct answer is option 1
Compounds which gives different ions in solution although they have same composition are called ionisation isomerism.
This form of isomerism arises when the counter ion in a complex salt is itself a potential ligand and can displace a ligand which can then become the counter ion.
An example is provided by the ionisation isomers [Co(NH3)4Br2]Cl and [Co(NH3)4BrCl]Br.
[Co(NH3)4Br2]Cl 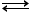 [Co(NH3)4Br2]+ + Cl-
[Co(NH3)4Br2]+ + Cl-
[Co(NH3)4BrCl]Br  [Co(NH3)4BrCl]+ + Br-
[Co(NH3)4BrCl]+ + Br-
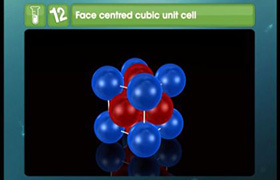
As given complex is octahedral it will also show the geometrical isomerism as follows:
Answered by Ramandeep | 19 Feb, 2020, 12:00: PM
Concept Videos
CBSE 12-science - Chemistry
Asked by prathyushagn1 | 09 Dec, 2020, 08:12: AM
CBSE 12-science - Chemistry
Asked by ABHILASHA | 31 Aug, 2020, 20:24: PM
CBSE 12-science - Chemistry
Asked by sha.bijoy17 | 07 Aug, 2020, 11:55: AM
CBSE 12-science - Chemistry
Asked by Shambhuhd79 | 22 Jun, 2020, 11:09: AM
CBSE 12-science - Chemistry
Asked by jain.pradeep | 19 Feb, 2020, 09:20: AM
CBSE 12-science - Chemistry
Asked by smit230503 | 04 Feb, 2020, 20:56: PM
CBSE 12-science - Chemistry
Asked by monishadubey202 | 08 Jan, 2020, 15:42: PM
CBSE 12-science - Chemistry
Asked by Chakshu29saini | 17 Sep, 2019, 18:19: PM
CBSE 12-science - Chemistry
Asked by bjayanta | 24 Mar, 2019, 20:56: PM
CBSE 12-science - Chemistry
Asked by himanshuneb | 28 Jan, 2019, 22:33: PM