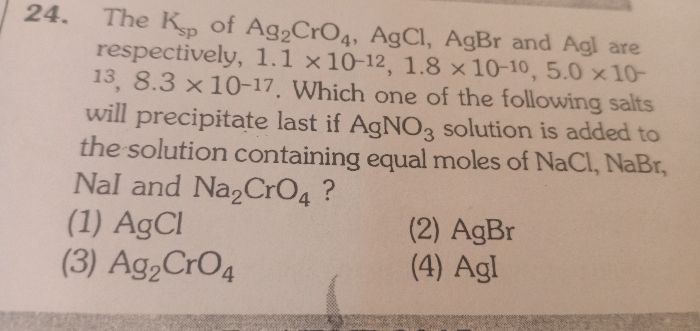CBSE Class 11-science Answered
Equal volumes of 0.1 M MgSO4 and 0.1 M Ba Cl2 are mixed together. Predict the formation of BaSO4 precipitate. Given Ksp of BaSO4 = 1.5 x 10-9
Asked by Topperlearning User | 16 Jun, 2016, 05:24: PM
BaSO4  Ba2+ + SO4-
Ba2+ + SO4-
 Ba2+ + SO4-
Ba2+ + SO4-Ksp = [Ba2+][ SO4-2] = 1.5 x 10-9
Concentration of[Ba2+]and [ SO4-2] in the final solution
[Ba2+] = 0.1 M/2 = 0.05 M
[ SO4-2] = 0.1 M/2 = 0.05 M
Now Ionic product = 0.05 x 0.05 = 2.5 x 10 -3 which is greater than Ksp = [Ba2+][ SO4-2] = 1.5 x 10-9
Hence the precipitation of BaSO4 will occur.
Answered by | 16 Jun, 2016, 07:24: PM
Concept Videos
CBSE 11-science - Chemistry
Asked by achamerahul2 | 21 Apr, 2020, 02:33: PM
CBSE 11-science - Chemistry
Asked by achamerahul2 | 21 Apr, 2020, 02:32: PM
CBSE 11-science - Chemistry
Asked by mufeedatvp2000 | 18 Apr, 2020, 02:21: PM
CBSE 11-science - Chemistry
Asked by Anish | 23 Aug, 2019, 01:48: AM
CBSE 11-science - Chemistry
Asked by jhajuhi19 | 12 Jun, 2019, 07:23: PM
CBSE 11-science - Chemistry
Asked by Prakash | 28 Jun, 2018, 06:09: PM
CBSE 11-science - Chemistry
Asked by gganga | 10 Apr, 2018, 06:31: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 04 May, 2015, 03:13: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 30 Apr, 2015, 02:30: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 16 Jun, 2016, 05:24: PM