ICSE Class 10 Answered
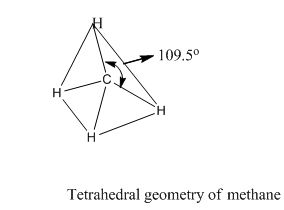
(1) Tetrahedral structure:
Methane: an example of tetrahedral geometry. The carbon has 4 valence electrons and thus needs 4 more electrons from four hydrogen atoms to complete its octet. The hydrogen atoms are as far apart as possible at 109o bond angle.
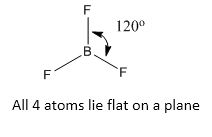
(2) Planar structure:
Boron trifluoride: an example of trigonal planner geometry. The boron has 3 valence electrons and these orbitals hybridized to sp2 and formed three same energy sp2 hybridized orbitals are coplanar and lying 120o apart, each containing one electron. These hybrid orbitals then overlap with the half-filled p-orbitals of three fluorine atoms forming three co-planer B-F electron pair bonds.
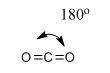
(3) Linear structure:
Carbon dioxide: It is an example of linear molecules. In this C is sp2 Hybridized to form hybrid orbitals which is overlapped by oxygen atom resulting in the formation of the double bond with linear geometry and 180o bond angle.