CBSE Class 11-science Answered
What is electropositive character? Why alkali metals form only monovalent ions?
Asked by Topperlearning User | 12 Aug, 2014, 13:43: PM
The electropositive character of an element is expressed in terms of the tendency of its atom to release electrons:
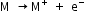
All the alkali metals are strongly electropositive or metallic in character, since they have low ionization energies and their atoms readily lose the valence electron. As the ionization energies decrease down the family, the electron releasing tendency or electropositive character is expected to increase down the family.
Answered by | 12 Aug, 2014, 15:43: PM
Concept Videos
CBSE 11-science - Chemistry
Asked by shreyasharma94.11dga | 23 Mar, 2022, 23:42: PM
CBSE 11-science - Chemistry
Asked by mrassam2711 | 15 Jan, 2021, 23:54: PM
CBSE 11-science - Chemistry
Asked by sg908580 | 21 Jan, 2020, 21:16: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 28 Jun, 2016, 13:37: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 28 Jun, 2016, 13:37: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 28 Jun, 2016, 13:35: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 04 Jun, 2014, 13:23: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 04 Jun, 2014, 13:23: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 12 Aug, 2014, 13:43: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 04 Jun, 2014, 13:23: PM




