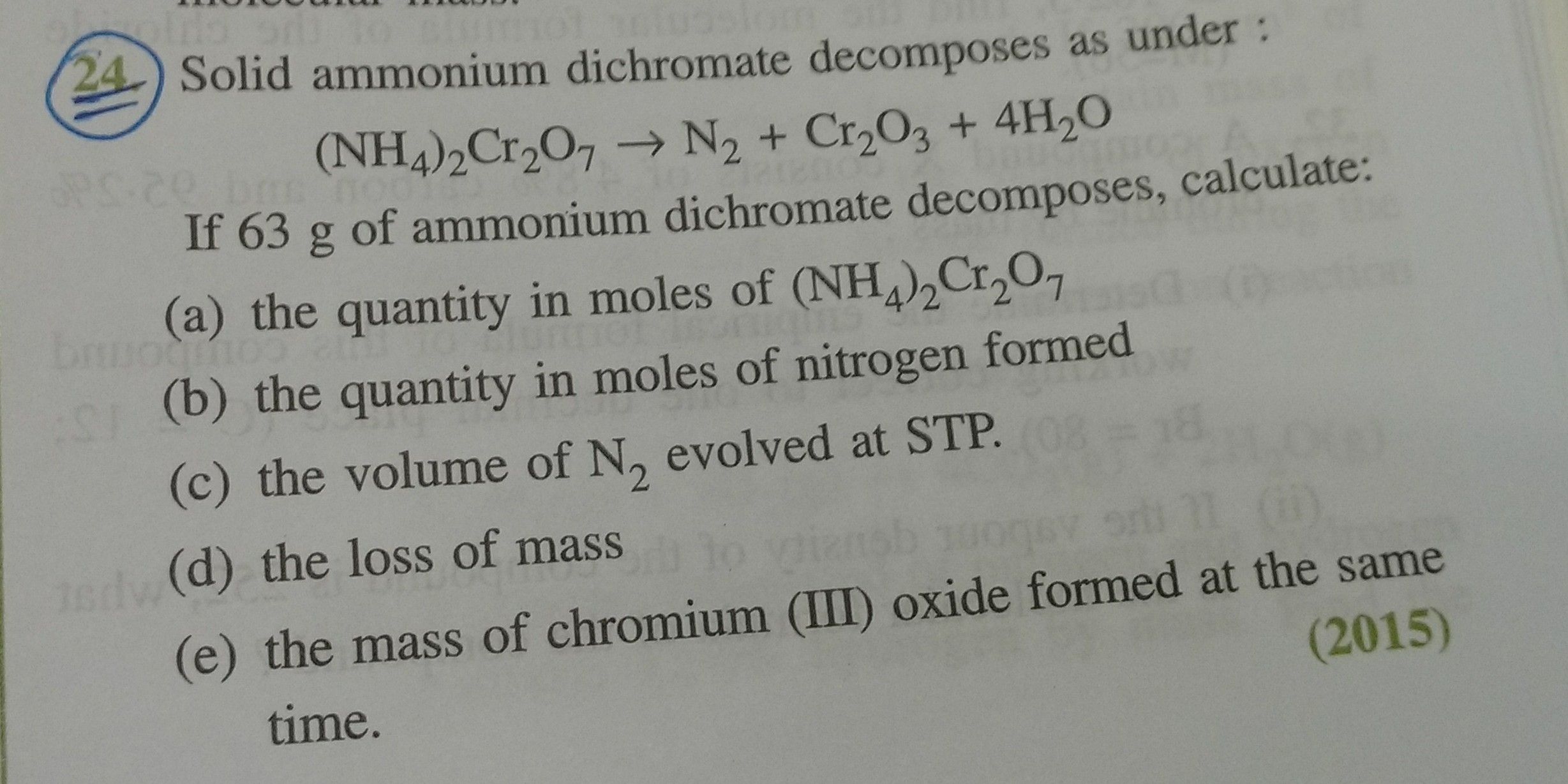ICSE Class 10 Answered
please solve the (v) part.

Asked by Kanwaranita10 | 16 Feb, 2020, 11:22: AM
option (B) is correct.
Given:
Mass of gas 'X' = 10 gm
Mass of H2 = 2 gm
We know,
Vapour density is given by,
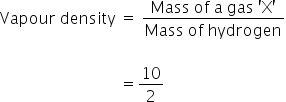
Vapour density = 5
Relative molecular mass of gas 'X' = Vapour density × 2
= 5 × 2 = 10
Answered by Varsha | 17 Feb, 2020, 11:13: AM
Application Videos
Concept Videos
ICSE 10 - Chemistry
Asked by maybe.kushagra | 25 Jan, 2024, 03:12: AM
ICSE 10 - Chemistry
Asked by srinu2020.ravipati | 16 Sep, 2020, 03:33: PM
ICSE 10 - Chemistry
Asked by Gurdev71 | 24 Jun, 2020, 12:41: PM
ICSE 10 - Chemistry
Asked by Kanwaranita10 | 16 Feb, 2020, 11:22: AM
ICSE 10 - Chemistry
Asked by aashimegh | 17 Aug, 2019, 02:24: PM
ICSE 10 - Chemistry
Asked by aashimegh | 03 Aug, 2019, 11:50: AM
ICSE 10 - Chemistry
Asked by Shrinivasdangi07 | 21 Mar, 2019, 10:34: PM
ICSE 10 - Chemistry
Asked by johncena9384 | 26 Oct, 2018, 04:08: PM
ICSE 10 - Chemistry
Asked by yajay0441 | 27 Aug, 2018, 03:15: PM