CBSE Class 11-science Answered
Please answer the following question.
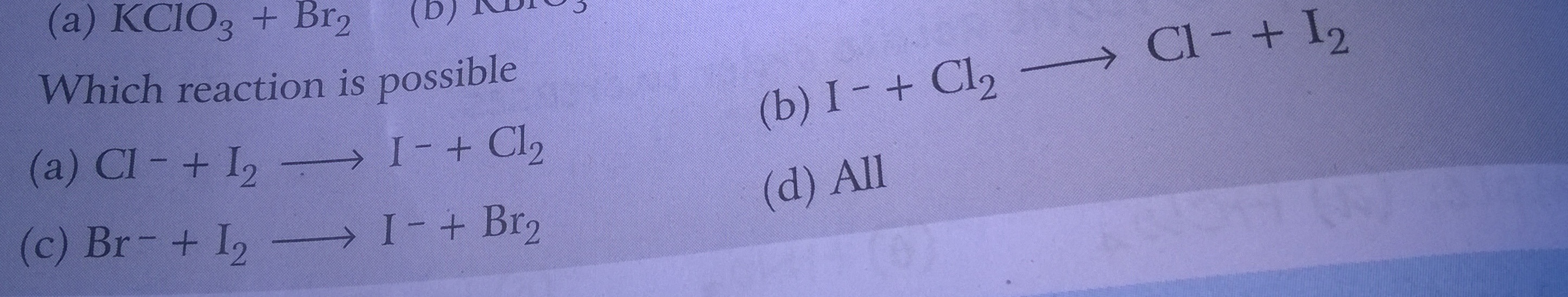
Asked by Balbir | 19 Jun, 2018, 18:36: PM
When there is a combination of Halogen (Cl2, Br2, I2) with a halide (Cl-, Br-, I-) then only stronger oxidising and strong reducing agents are combined.
Halogens are oxidising agents with the order: Cl2> Br2>I2
whereas halides are reducing agents: Cl-< Br-< I-
In option 1:
Cl- + I2 → I- + Cl2
In above reaction, Cl- is a weak R.A and I2 is also a weak O.A. HENCE NOT POSSIBLE
In option 3:
Br- + I2 → I- + Br2
In above reaction, Br- is a moderate R.A but I2 is a weak O.A. HENCE NOT POSSIBLE
In option 2:
I- + Cl2 → Cl- + I2
In above reaction, I- is a strong R.A. and Cl2 is also a strong O.A. HENCE THIS REACTION IS POSSIBLE
Answered by Ramandeep | 21 Jun, 2018, 12:27: PM
Concept Videos
CBSE 11-science - Chemistry
Asked by patrasudipkumar191 | 05 Jul, 2024, 09:36: AM
CBSE 11-science - Chemistry
Asked by jaip83491 | 24 Jan, 2021, 10:43: AM
CBSE 11-science - Chemistry
Asked by sulaikhasulu393 | 07 Jun, 2020, 22:51: PM
CBSE 11-science - Chemistry
Asked by debjit_dm | 04 May, 2020, 15:08: PM
CBSE 11-science - Chemistry
Asked by Balbir | 30 Jun, 2019, 15:45: PM
CBSE 11-science - Chemistry
Asked by govtsecschoolnayaganv051 | 05 Sep, 2018, 13:32: PM
CBSE 11-science - Chemistry
Asked by govtsecschoolnayaganv051 | 09 Aug, 2018, 16:52: PM
CBSE 11-science - Chemistry
Asked by dineshchem108 | 17 Jul, 2018, 18:02: PM
CBSE 11-science - Chemistry
Asked by vaagai2353 | 29 Jun, 2018, 21:55: PM






