CBSE Class 11-science Answered
NaH+H2O=NaOH+ H2 which type of redox reaction is this? Pls explain in detail
Asked by govtsecschoolnayaganv051 | 09 Aug, 2018, 16:52: PM
NaH+H2O → NaOH+ H2 is a type of displacement redox reaction.
Explanation:
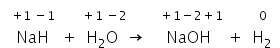
In the above reaction, Hydrogen of NaH is changing its oxidation state from -1 to +1 and Hydrogen from water changes from +1 to 0.
Hence NaH is reducing agent and H2O is the oxidizing agent.
Answered by Ramandeep | 13 Aug, 2018, 11:55: AM
Concept Videos
CBSE 11-science - Chemistry
Asked by patrasudipkumar191 | 05 Jul, 2024, 09:36: AM
CBSE 11-science - Chemistry
Asked by jaip83491 | 24 Jan, 2021, 10:43: AM
CBSE 11-science - Chemistry
Asked by sulaikhasulu393 | 07 Jun, 2020, 22:51: PM
CBSE 11-science - Chemistry
Asked by debjit_dm | 04 May, 2020, 15:08: PM
CBSE 11-science - Chemistry
Asked by Balbir | 30 Jun, 2019, 15:45: PM
CBSE 11-science - Chemistry
Asked by govtsecschoolnayaganv051 | 05 Sep, 2018, 13:32: PM
CBSE 11-science - Chemistry
Asked by govtsecschoolnayaganv051 | 09 Aug, 2018, 16:52: PM
CBSE 11-science - Chemistry
Asked by dineshchem108 | 17 Jul, 2018, 18:02: PM
CBSE 11-science - Chemistry
Asked by vaagai2353 | 29 Jun, 2018, 21:55: PM






