CBSE Class 11-science Answered
nitrogen filled in 1 kg cylinder @150 bar . then strored it at 0 degree cetrigrade. for 24 hrs. what will be changes in properties of nitrogen.
Asked by design1.bharatifire | 21 Jun, 2019, 18:35: PM
At this high pressutre and low temperature, intermolecular forces in nitrogen is increased and liquification takes place. So nitrogen will be converted in liquid from its gaseous state.
Answered by Ravi | 24 Jun, 2019, 12:58: PM
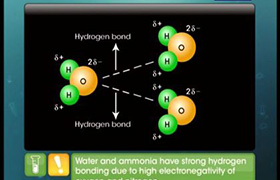
Concept Videos
CBSE 11-science - Chemistry
Asked by neerudhawanbpsmv | 28 Jun, 2021, 09:22: AM
CBSE 11-science - Chemistry
Asked by swatipuspapatel | 10 Jun, 2020, 18:19: PM
CBSE 11-science - Chemistry
Asked by vanyashree106 | 13 Dec, 2019, 21:38: PM
CBSE 11-science - Chemistry
Asked by Asif.k1992 | 12 Sep, 2019, 14:41: PM
CBSE 11-science - Chemistry
Asked by ajitbhot73 | 25 Aug, 2019, 11:52: AM
CBSE 11-science - Chemistry
Asked by design1.bharatifire | 21 Jun, 2019, 18:35: PM
CBSE 11-science - Chemistry
Asked by anitabahuguna | 04 May, 2019, 21:57: PM
CBSE 11-science - Chemistry
Asked by Jatinaa786 | 19 Sep, 2018, 02:53: AM
CBSE 11-science - Chemistry
Asked by arjunshrivastav668 | 17 Mar, 2018, 10:21: AM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 04 Jun, 2014, 13:23: PM