CBSE Class 11-science Answered
Gases exert pressure on the walls of the container why? Explain in one two answer
Asked by vanyashree106 | 13 Dec, 2019, 21:38: PM
Molecules of gases are continuously colliding with each other and with the walls of the container. When molecule colloids with the wall, they exert small force on the wall i.e., the pressure. The pressure exerted by the gas is due to the sum of all these collision forces. The more particles that hit the walls, the pressure will increase.
Answered by Ramandeep | 16 Dec, 2019, 11:52: AM
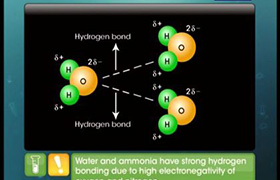
Concept Videos
CBSE 11-science - Chemistry
Asked by neerudhawanbpsmv | 28 Jun, 2021, 09:22: AM
CBSE 11-science - Chemistry
Asked by swatipuspapatel | 10 Jun, 2020, 18:19: PM
CBSE 11-science - Chemistry
Asked by vanyashree106 | 13 Dec, 2019, 21:38: PM
CBSE 11-science - Chemistry
Asked by Asif.k1992 | 12 Sep, 2019, 14:41: PM
CBSE 11-science - Chemistry
Asked by ajitbhot73 | 25 Aug, 2019, 11:52: AM
CBSE 11-science - Chemistry
Asked by design1.bharatifire | 21 Jun, 2019, 18:35: PM
CBSE 11-science - Chemistry
Asked by anitabahuguna | 04 May, 2019, 21:57: PM
CBSE 11-science - Chemistry
Asked by Jatinaa786 | 19 Sep, 2018, 02:53: AM
CBSE 11-science - Chemistry
Asked by arjunshrivastav668 | 17 Mar, 2018, 10:21: AM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 04 Jun, 2014, 13:23: PM