CBSE Class 12-science Answered
Dear Student,
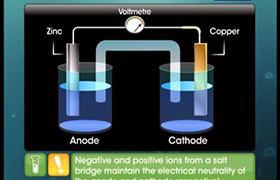
Electrochemical cell representation (IUPAC conventions)
Let us illustrate the convention taking the example of Daniel cell.
(a) Anodic half-cell is written on left and cathodic half-cell on right hand side.
Zn(s) |ZnSO4 (sol)||CuSO4 (sol)|Cu(s)
(b) Two half cells are separated by double vertical lines: Double vertical lines indicate slat bridge or any type of porous partition.
(c) EMF (electromotive force) may be written on the right hand side of the cell.
(d)Single vertical lines indicate the phase separation between electrode and electrolyte solution.
Zn|Zn2+|| Cu2+|Cu
(e) Invert eletrodes are represented in the bracket
Zn|ZnSO4 || H+| H2, Pt
EMF of cell should be positive otherwise it will not be feasible in the given direction.
Zn|ZnSO4||CuSO4|Cu
E = + 1.10 volt (Feasible)
Cu|CuSO4||ZnSO4|Zn
E = – 1.10 volt (Not Feasible)