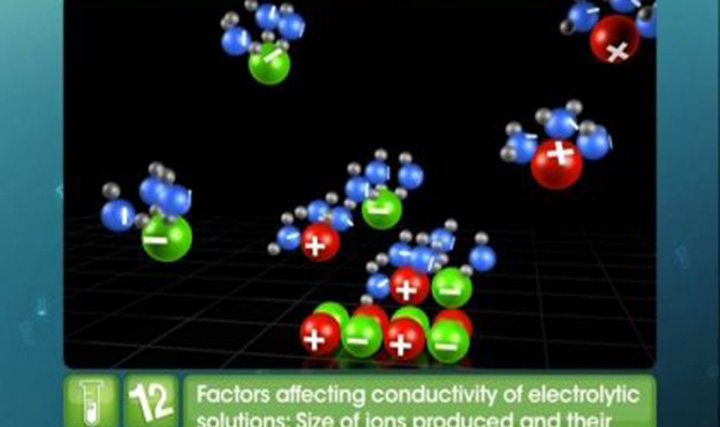
CBSE Class 12-science - Electrochemical Cells Videos
Redox Reactions and Galvanic Cells
This video explains meaning of Redox reaction, construction and working of Galvanic cell and Salt Bridge.
More videos from this chapter
View All-
consider the following reaction

- what is electrochemical cell
- what is galvanic cell
- the electric charge present on di positive magnesium ion is
- How to make simple equations for anode and cathode in standard electrode potential? How to understand their sign if the value of reduction and oxidation in questions?
- in an electrochemical cell
- Calculate the no. Of coulpmb required to deposit 40.5g AL when the electrode rxn is AL3+ +3e--->AL
- when zinc plate is dipped into a blue coloured solution of cuso4 then its colour becomes white why
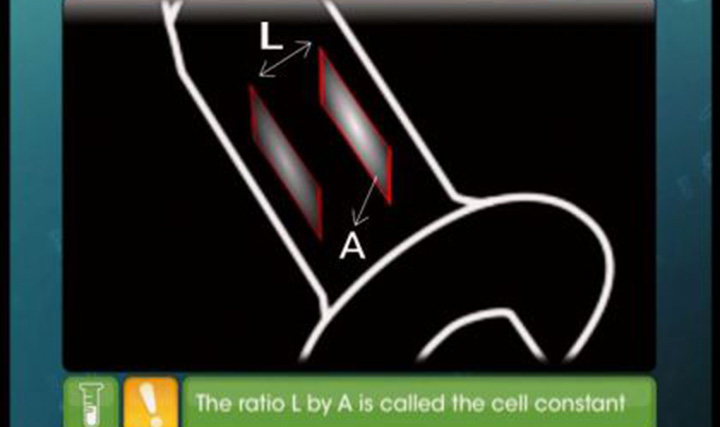
- cell constant G*= l /A Here A stands for area of Electrode or Electrolytic solution
- What is galvanic cell?