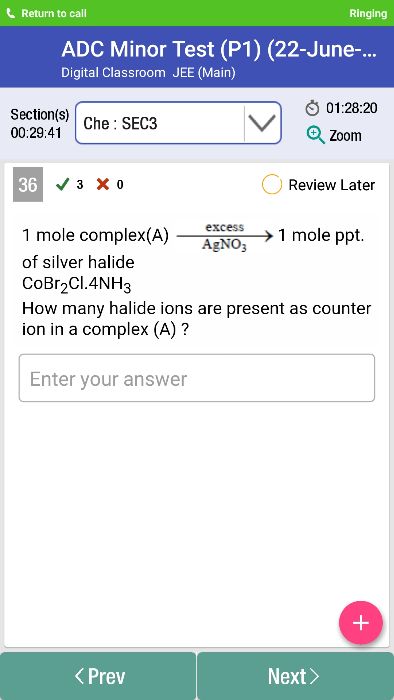
CBSE Class 12-science Answered
How to calculate primary valence
Asked by Chakshu29saini | 17 Sep, 2019, 06:19: PM
According to Werner's Theory in coordination compounds oxidation state of central metal is called as it's primary valency.
For example,In complex [CoCl2(NH3)4]Cl
Oxidation state of Co=+3
So Primary valency=+3
Answered by Ravi | 17 Sep, 2019, 07:10: PM
Concept Videos
CBSE 12-science - Chemistry
Asked by prathyushagn1 | 09 Dec, 2020, 08:12: AM
CBSE 12-science - Chemistry
Asked by ABHILASHA | 31 Aug, 2020, 08:24: PM
CBSE 12-science - Chemistry
Asked by sha.bijoy17 | 07 Aug, 2020, 11:55: AM
CBSE 12-science - Chemistry
Asked by Shambhuhd79 | 22 Jun, 2020, 11:09: AM
CBSE 12-science - Chemistry
Asked by jain.pradeep | 19 Feb, 2020, 09:20: AM
CBSE 12-science - Chemistry
Asked by smit230503 | 04 Feb, 2020, 08:56: PM
CBSE 12-science - Chemistry
Asked by monishadubey202 | 08 Jan, 2020, 03:42: PM
CBSE 12-science - Chemistry
Asked by Chakshu29saini | 17 Sep, 2019, 06:19: PM
CBSE 12-science - Chemistry
Asked by bjayanta | 24 Mar, 2019, 08:56: PM
CBSE 12-science - Chemistry
Asked by himanshuneb | 28 Jan, 2019, 10:33: PM