CBSE Class 12-science Answered
explain
Asked by | 28 Mar, 2008, 01:01: AM
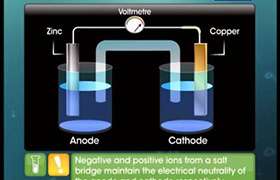
This is explained by Nernst. He showed in Daniel cell that E(cell) depends on the concentration of both Cu+2 & Zn+2 ions. It increases with increase in conc of Cu ions and decrease in conc of Zn ions.
E(cell) = E 0(cell) - RT/nF ln (C)c ( D)d / (A) a (B)b.
Answered by | 21 Dec, 2017, 12:33: PM
Concept Videos
CBSE 12-science - Chemistry
Asked by summiafroz31 | 06 Feb, 2024, 08:39: PM
CBSE 12-science - Chemistry
Asked by aryamankrsinha2002 | 29 Nov, 2023, 11:39: AM
CBSE 12-science - Chemistry
Asked by banneramadevi | 26 Jul, 2023, 08:51: PM
CBSE 12-science - Chemistry
Asked by Poojanisha1988 | 19 Jul, 2023, 09:59: PM
CBSE 12-science - Chemistry
Asked by jajimuji2306 | 03 Apr, 2022, 01:38: PM
CBSE 12-science - Chemistry
Asked by Harshfarwaha | 23 Jul, 2020, 03:27: PM
CBSE 12-science - Chemistry
Asked by sourabhkumar9923 | 19 May, 2020, 08:21: PM
CBSE 12-science - Chemistry
Asked by ssharondaniel | 27 Jul, 2019, 06:22: PM
CBSE 12-science - Chemistry
Asked by kripanjalihimansu | 28 Feb, 2019, 06:57: AM
CBSE 12-science - Chemistry
Asked by govtsecschoolnayaganv051 | 12 Sep, 2018, 05:40: PM