CBSE Class 11-science Answered
Explain the structure of Diborane and banana bonds?
Asked by | 23 Feb, 2013, 07:24: AM
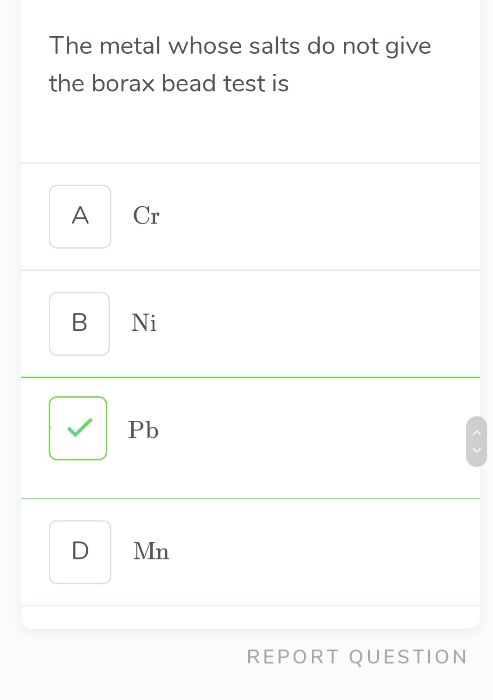
In diborane four terminal B-H bonds are two centre - two electron bonds. Two bridge B ? H ? B bonds (or banana bonds) are three centre - two electron bonds (shown in figure).
.png)
2 boron and 4 terminal hydrogen atoms (Ht) lie in one plane, while the other two bridging hydrogen atoms (Hb) lie in a plane perpendicular to the plane of boron atoms. Again, of the two bridging hydrogen atoms, one H atom lies above the plane and the other lies below the plane. The terminal bonds are regular two-centre two-electron (2c 2e) bonds, while the two bridging (BHB) bonds are three-centre two-electron (3c 2e) bonds.
Answered by | 23 Feb, 2013, 05:15: PM
Concept Videos
CBSE 11-science - Chemistry
Asked by singhnikita8717 | 05 Aug, 2021, 10:44: AM
CBSE 11-science - Chemistry
Asked by jaswindernkd | 21 Jun, 2020, 05:04: PM
CBSE 11-science - Chemistry
Asked by mufeedatvp2000 | 18 Apr, 2020, 02:21: PM
CBSE 11-science - Chemistry
Asked by Molaypaul700 | 10 Feb, 2020, 10:32: PM
CBSE 11-science - Chemistry
Asked by ABHILASHA | 08 Sep, 2019, 06:46: PM
CBSE 11-science - Chemistry
Asked by krishdabhoya2003 | 05 Aug, 2019, 08:19: AM
CBSE 11-science - Chemistry
Asked by vishakhachandan026 | 12 Jun, 2019, 09:20: AM
CBSE 11-science - Chemistry
Asked by pb_ckt | 28 Apr, 2019, 01:40: PM
CBSE 11-science - Chemistry
Asked by satya785583 | 16 Mar, 2019, 09:18: PM
CBSE 11-science - Chemistry
Asked by govtsecschoolnayaganv051 | 03 Jan, 2019, 01:09: PM