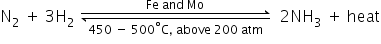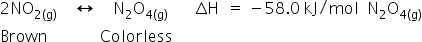CBSE Class 11-science Answered
plzzzz solve

Asked by satya785583 | 16 Mar, 2019, 09:18: PM
(a)
(i)
(ii)
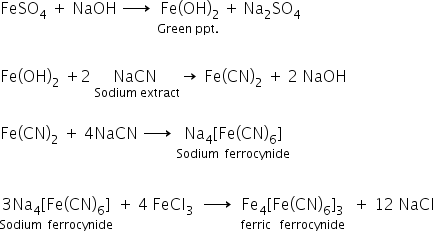
(b) Nitrogen, sulphur, halogens and phosphorus present in an organic compound are detected by Lassaignes test.
The elements present in the compound are converted from covalent form to ionic form by fusing the compound with sodium metal.
(c) An aqueous solution of CuSO4 cannot be stored in a container made by zinc because zinc is more active than copper therefore, zinc will displace copper from its salt solution.Also, the reduction potential of copper is less as compared to zinc. hence an aqueous solution of CuSO4 will reduce itself into copper and zinc sulphate will be formed.The reaction occurs as follows:
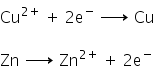
Answered by Ramandeep | 19 Mar, 2019, 03:37: PM
Concept Videos
CBSE 11-science - Chemistry
Asked by ABHILASHA | 08 Sep, 2019, 06:46: PM
CBSE 11-science - Chemistry
Asked by vishakhachandan026 | 12 Jun, 2019, 09:20: AM
CBSE 11-science - Chemistry
Asked by pb_ckt | 28 Apr, 2019, 01:40: PM
CBSE 11-science - Chemistry
Asked by satya785583 | 16 Mar, 2019, 09:18: PM
CBSE 11-science - Chemistry
Asked by govtsecschoolnayaganv051 | 03 Jan, 2019, 01:09: PM
CBSE 11-science - Chemistry
Asked by govtsecschoolnayaganv051 | 03 Jan, 2019, 12:34: PM
CBSE 11-science - Chemistry
Asked by anushkamoudgil123 | 02 Nov, 2018, 05:47: AM
CBSE 11-science - Chemistry
Asked by arunavamitra50 | 04 Jul, 2018, 09:11: PM
CBSE 11-science - Chemistry
Asked by arunavamitra50 | 04 Jul, 2018, 09:10: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 31 May, 2016, 01:10: PM