CBSE Class 11-science Answered
Does shielding effect occur between atomic orbitals of the same quantum number? Explain with reasons
Asked by fishtailfever | 08 Sep, 2019, 23:00: PM
When we are talking about the same shell, electron experiences a force of repulsion due to the presence of other electrons in the same shell. This force pushes newly added electron slightly apart but since effective nuclear charge in the outermost orbit is increased, the nucleus pulls it with greater force. so force in same quantum number orbitals is negligible as compared to other repulsion and attraction.
Answered by Ravi | 10 Sep, 2019, 16:06: PM
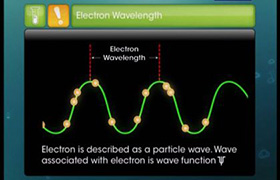
Concept Videos
CBSE 11-science - Chemistry
Asked by o230397 | 23 Sep, 2023, 14:48: PM
CBSE 11-science - Chemistry
Asked by ks1221516 | 14 Nov, 2021, 18:46: PM
CBSE 11-science - Chemistry
Does shielding effect occur between atomic orbitals of the same quantum number? Explain with reasons
Asked by fishtailfever | 08 Sep, 2019, 23:00: PM
CBSE 11-science - Chemistry
Asked by patra04011965 | 09 Aug, 2019, 17:19: PM
CBSE 11-science - Chemistry
Asked by bhrjitu | 21 Jul, 2019, 08:08: AM
CBSE 11-science - Chemistry
Asked by pb_ckt | 06 Jun, 2019, 09:15: AM
CBSE 11-science - Chemistry
Asked by shahrithik07 | 18 Oct, 2018, 17:26: PM
CBSE 11-science - Chemistry
Asked by chinjalsoni911 | 16 Oct, 2018, 18:14: PM
CBSE 11-science - Chemistry
Asked by arunavamitra50 | 18 Jun, 2018, 18:39: PM
CBSE 11-science - Chemistry
Asked by badalsharma9929 | 13 Jun, 2018, 08:43: AM