CBSE Class 11-science Answered
Calculate the pH of a solution that has a [OH1-] = 1 X 10-5 M
Asked by Topperlearning User | 16 Jun, 2016, 17:21: PM
First determine pOH = -log[OH- ] = -log [1 X 10-5 ] = 5
Now we know, Ionic product of water = pH + pOH = 14
pH + 5 = 14 Or, pH = 14 -5 = 9
Answered by | 16 Jun, 2016, 19:21: PM
Concept Videos
CBSE 11-science - Chemistry
Asked by Topperlearning User | 16 Jun, 2016, 17:22: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 29 Apr, 2015, 14:41: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 30 Apr, 2015, 08:49: AM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 30 Apr, 2015, 09:57: AM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 30 Apr, 2015, 10:51: AM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 30 Apr, 2015, 11:04: AM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 30 Apr, 2015, 11:08: AM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 16 Jun, 2016, 17:21: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 16 Jun, 2016, 17:21: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 16 Jun, 2016, 17:21: PM




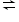 H+ + HS-
H+ + HS-