CBSE Class 12-science Answered
sir/madam
Asked by Nevin | 10 Mar, 2013, 06:29: AM
(i) Phenol reacts with ethanoyl chloride at room temperature, although the reaction isn't as fast as the one between ethanoyl chloride and an alcohol. Phenyl ethanoate is formed together with hydrogen chloride gas.
![]()

(ii) Ethanol is oxidised
- with acidified Potassium Dichromate, K2Cr2O7, or
- with acidified Sodium Dichromate, Na2Cr2O7, or
- with acidified potassium permanganate, KMnO4,
[O] C2H5OH ==> CH3CHO + H2O Ethanol Ethanal
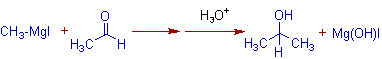
The addition of Methylmagnesium iodide to acetaldehyde gives Isopropyl alcohol.
Answered by | 10 Mar, 2013, 10:02: AM
Application Videos
Concept Videos
CBSE 12-science - Chemistry
Asked by guneetk44 | 28 Apr, 2024, 10:39: AM
CBSE 12-science - Chemistry
Asked by kavitabawane190 | 08 Mar, 2024, 05:24: PM
CBSE 12-science - Chemistry
Asked by rp0055293 | 07 Feb, 2024, 08:28: AM
CBSE 12-science - Chemistry
Asked by sanyaagrahari8888 | 28 Nov, 2023, 01:07: PM
CBSE 12-science - Chemistry
Asked by bithikapurkait09052005 | 24 Jun, 2022, 08:35: AM
CBSE 12-science - Chemistry
Asked by rohanm | 14 Feb, 2022, 04:49: PM
CBSE 12-science - Chemistry
Asked by vipulverma | 14 Feb, 2022, 04:44: PM
CBSE 12-science - Chemistry
Asked by kaziryan.05 | 23 Jun, 2021, 08:02: PM
CBSE 12-science - Chemistry
Asked by deepakatur454 | 28 Nov, 2020, 04:29: PM
CBSE 12-science - Chemistry
Asked by holebasubudihal | 09 Aug, 2020, 02:58: PM










