CBSE Class 12-science Answered
Show in case of body centered cube only 68% the space is occupied and rest is unoccupied.
Asked by Topperlearning User | 16 Jun, 2016, 03:54: PM
Atom in the body diagonal will be in touch with other two atoms diagonally.
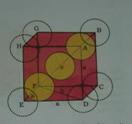
In Δ EFD ,
b2 = a2 +a2= 2a2,
b= √2a
Now in ΔAFD
c2 = a2+b2 = a2 + 2a2 = 3a2
c = √3a
√3a = 4r
r = √3/4 x a
Volume of the cube = a3 will be equal to a3 = [4/√3 x r]3.
Packing efficiency = volume occupied by two spheres in the unit cell x 100 / total volume of the unit cell %
= 2 x (4/3) Πr3 x 100/ [(4/√3)r]3 %
=(8/3) Πr3 x100 / 64/(3√3)r3 % = 68%.
Answered by | 16 Jun, 2016, 05:54: PM
Concept Videos
CBSE 12-science - Chemistry
Asked by rasmimajhi07 | 21 Nov, 2023, 10:22: PM
CBSE 12-science - Chemistry
Asked by pradeepkumar70258 | 04 Oct, 2023, 10:30: PM
CBSE 12-science - Chemistry
Asked by akash9322793205 | 12 Aug, 2021, 09:30: AM
CBSE 12-science - Chemistry
Asked by thakurranjan54 | 08 Feb, 2021, 06:32: PM
CBSE 12-science - Chemistry
Asked by sildasholly2002 | 26 Jul, 2020, 06:37: PM
CBSE 12-science - Chemistry
Asked by tejuaaygole | 11 Jul, 2020, 10:40: AM
CBSE 12-science - Chemistry
Asked by chandlerbong164 | 12 Apr, 2020, 07:22: PM
CBSE 12-science - Chemistry
Asked by mangeshkale423 | 25 Feb, 2020, 05:12: PM
CBSE 12-science - Chemistry
Asked by upasana.sobti | 16 Jan, 2019, 01:59: PM




