CBSE Class 12-science Answered
Molar conductance values at infinite dilution of Na+ and Cl- ions are 50.11*10^-4Sm^2mol^-1 and 76.34*10^-4Sm^2mol^-1 respectively. Calculate the transport number of Na+ ion.
Asked by mrudulmahadev1311 | 22 Aug, 2019, 07:47: AM
Given:
Molar conductance at infinite dilution;
λ+Na = 50.11×10−4 Sm2mol−1
λ+Cl = 76.34×10−4 Sm2mol−1
Molar conductance of NaCl is = λ+Na + λ+Cl
= 50.11×10−4 + 76.34×10−4
= 126.45×10−4 Sm2mol−1
Transport number of Na+
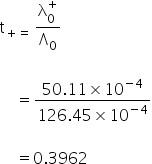
Answered by Varsha | 22 Aug, 2019, 12:32: PM
Concept Videos
CBSE 12-science - Chemistry
Asked by samskruthikrishn | 12 Jan, 2024, 10:11: AM
CBSE 12-science - Chemistry
Asked by 2507king2006 | 03 Oct, 2023, 07:12: AM
CBSE 12-science - Chemistry
Asked by eyemunshah | 20 Nov, 2021, 11:19: PM
CBSE 12-science - Chemistry
Asked by samisiddiqui013 | 29 May, 2021, 07:49: PM
CBSE 12-science - Chemistry
Asked by mrudulmahadev1311 | 22 Aug, 2019, 07:47: AM
CBSE 12-science - Chemistry
Asked by mrudulmahadev1311 | 21 Aug, 2019, 12:17: PM
CBSE 12-science - Chemistry
Asked by rakeshraghav33 | 26 May, 2018, 05:06: PM
CBSE 12-science - Chemistry
Asked by Topperlearning User | 04 Jun, 2014, 01:23: PM
CBSE 12-science - Chemistry
Asked by Topperlearning User | 16 Jun, 2014, 10:56: AM