CBSE Class 11-science Answered
explain why pressure of CO2 becomes constant when equilibrium is attained in the decomposition of CaCO3 in a closed vessel.
Asked by ABHILASHA | 27 Oct, 2019, 12:26: AM
According to Le Chatelier's principle of Chemical-equilibrium, At equilibirium state, When pressure increases then reaction shifts in the direction where number of moles are less and When pressure decreases then reaction shifts in the direction of higher number of moles.
In case of decomposition of CaCO3 -

In closed vessel, there is no chaange in number of moles, So pressure will remain constant at equilibrium.
Answered by Ravi | 30 Oct, 2019, 04:08: PM
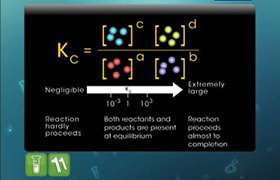
Concept Videos
CBSE 11-science - Chemistry
Asked by visank90 | 24 Nov, 2023, 10:45: AM
CBSE 11-science - Chemistry
Asked by rakhikumarithakur4 | 22 May, 2020, 08:21: PM
CBSE 11-science - Chemistry
Asked by arshrana3272 | 04 Mar, 2020, 02:57: PM
CBSE 11-science - Chemistry
Asked by hsdhall.2005 | 12 Nov, 2019, 11:40: PM
CBSE 11-science - Chemistry
Asked by govtsecschoolnayaganv051 | 19 Jul, 2018, 08:52: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 16 Jun, 2016, 05:17: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 16 Jun, 2016, 05:17: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 27 Apr, 2015, 03:08: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 27 Apr, 2015, 04:12: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 27 Apr, 2015, 04:15: PM




 6H2O (g) + 2N2 (g) How will the equilibrium shift if: a) The volume is increased, b) Helium gas is added
6H2O (g) + 2N2 (g) How will the equilibrium shift if: a) The volume is increased, b) Helium gas is added